Tox-Blog #8: Aluminum. Toxicity, risks, and the consequences for food and vaccine safety.
I love hot topics, and the toxicity of aluminum plays a central role in several battles around industrial food and vaccines. The perfect opportunity for me to jump into another debate.
You may also like:
Tox-Blog #1: Organic food | Tox-Blog #2: Glyphosate
Tox-Blog #3: Cinnamon | Tox-Blog #4: Aflatoxins
Tox-Blog #5: Coprine | Tox-Blog #6: Magic Mushrooms
Tox-Blog #7: Basic concepts of risk assessment
After clarifying some basic concepts behind risk assessment (I recommend reading that post before diving into this one), let’s get serious now.

from pixabay
Intro: What is Aluminum?
Aluminium (chemical symbol Al) is a light metal that is found in (and obtained from) various minerals. Its unique physical and chemical properties have led to its widespread use in a large variety of different goods, tools, and other applications, including food additives and vaccinations.
Over the last decades, its toxicity has become more and more explored, and so nowadays there's a high level of anxiety and insecurity in our society on how to use it and what it might actually cause.
And indeed, this is an important topic. Let’s dive straight into it.
Aluminum is not Aluminum.
The first important lesson here is that the toxicity of Aluminum largely depends on in which form you take it up.
Humans get in contact with Aluminum either in its form as a metal (e.g. aluminum foil, dishes, etc.) or as an ion (Al3+, e.g. as food additive, food contaminant or in vaccines). Eating great amounts of the metal is a very unlikely case, so let’s focus on the ion.
Ions pair up, which is then called a “salt” – but do not confuse it with “the” salt (sodium chloride) please.
Now again, Al salt is not Al salt. The toxicological problem is the Al ion, that can only be taken up by our body if it is solved first. So depending on the solubility of the Al salt, we take up different amounts of Al ions. So on one hand, if we ingest something that solves quite well in water like aluminum citrate, our body takes up a lot of toxic Al ions (the same is valid for vaccinations, where the Al is already solved), but a mineral like aluminum silicate, which is almost completely insoluble, is quite harmless for the human body.
Al-containing food additives, for example Al silicate which is used as anti-caking agent, are generally insoluble in water and also acid-persistent, so they are considered non-toxic. Of course there are also lawful limits to be respected, calculated from TDI/TWI values. In general: not a problem.
However, Al can be a problematic food contaminant.
Imagine you are barbecuing, and you have those new fancy barbecue dishes for grilling vegetables, because you were told broiling them above the open flame is bad. The dishes are normally made with Aluminum metal. You put your vegetables into the dish, then you add some salt, pepper, herbs, oil. And in the end you add some lemon, because you like it, and it is healthy, yes?.
BAD DECISION. The lemon is very acidic (because of the citric acid), you heat the whole thing, so you have a hot acid in aluminum. The hot acid will oxidize some of the metallic aluminum, and solve the generated Al ions – you gain a solution of aluminum citrate, which is soluble and in turn will be taken up by your body.
But not only lemons are acidic. Similar contaminations can happen e.g. when cooking coffee with aluminum capsules (I’m looking at you, Nespresso!) etc.

Damn you, Nespresso! From pixabay.
And – to a certain extent – modern food processing often involves machineries containing aluminum, so there’s also the possibility to contaminate food via this way. According to the European Food Safety Authority, “foods with very high mean concentrations include tea leaves, herbs, cocoa and cocoa products, and spice” (Source)
Acute and chronic toxicity of aluminum
Acutely, high doses of Al can lead to lung/liver/kidney damages (depending on the way of uptake) and ultimately, to death. Relax, that is very unlikely to happen, unless you are witness to an industrial accident in a chemical plant. Food? No way.
Chronic toxicity is a much greater problem in our civilization. When taken up over a long time at elevated levels, Al can accumulate in bones and nerve tissue, and is suspected to cause or at least accelerate neurodegenerative diseases, in particular Alzheimer’s , and especially in persons with impaired kidney functions. (Source)
While the fact that Al is able to do this is more or less out of doubt, we are not 100% sure how it causes neural damages. One contributing pathway is the induction of oxidative stress, which in turn damages the tissue where the ions accumulated.
Based on the large amount of scientific data, the EFSA has defined a TWI of 1mg Al per kg bodyweight, which is considered safe, but also estimated that large parts of the European population are currently exceeding this limit with their lifestyle.
So yes, food contamination with Al (but not the use of its insoluble salts) is a problem for real.
Aluminum in Vaccines
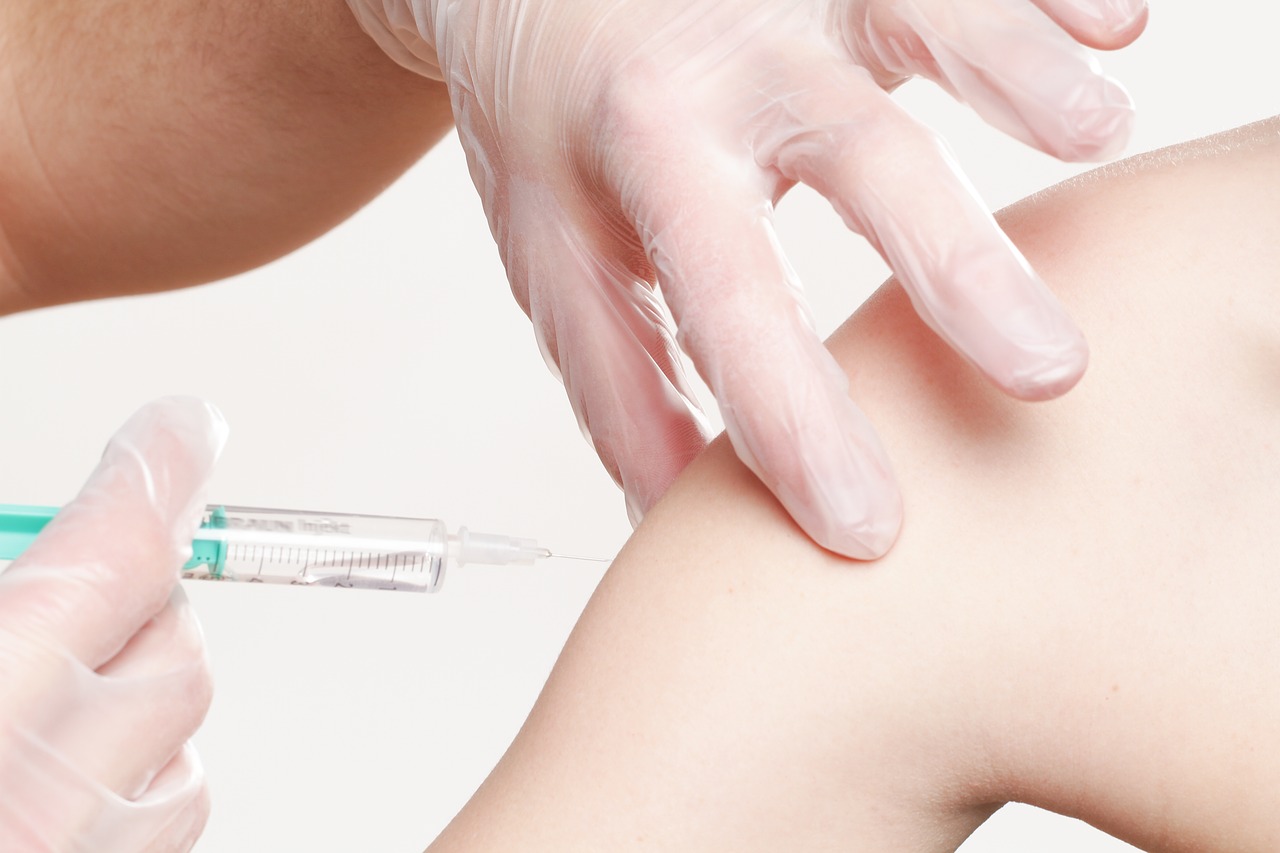
Yes, I do hate getting stung too, and I even more hate to watch my boy being stung. No reason to deny the usefulness for humanity, though. From pixabay
Now it gets interesting.
As every vaccination-sceptic person will be able to tell you, the neurotoxin aluminum is used in vaccines. And this is true, it serves as an adjuvant there.
As they can tell you as well, it is allowed in vaccines up to a doses that exceed the safety limits in food.
This is also - somewhat - true. The maximum amount of aluminum in a vaccine shot is 1.25 mg (FDA). Assuming a vaccinated baby has at least 2 kg bodyweight, this would be ~0.8 mg/kg BW.
While the TWI for adults is 1 mg/kg, there is an extra safety factor of 10 in place for infants, so for them, the TWI is 0.1 mg/kg.
So yes, even as most vaccines use significantly less aluminum, the theoretically allowed amount of Al ions in a vaccine shot is up to 8 times higher than the safety limit for infants in food.
So it’s true!!! They are poisoning our kids!!! Autism, neurologic damages, stupidity, aaaaaah!!!
No.
The TWI assesses the chronic toxicity, it means if you take up more than 1 mg/kg BW (or 0.1 mg/kg for infants) per week for a long time, you are at a somewhat theoretical risk (there's still a security factor of 100-1000 to overcome until you reach the first LOAELs).
But getting one shot of Al-containing vaccine is not a chronic, but an acute exposure!
And the toxicological thresholds for acute Al toxicity are much, much higher. Even with all the different vaccines that kids routinely get these days, they take up much less of total Al by vaccination than by food, breastmilk, and inhaling fumes during their first years (source). This is why the large majority of scientists in the field thinks Al in vaccines is safe.
This, and the very extensive studies that have been performed on the safety of vaccines, of course.
But for the sake of scientific correctness, there is one exception: If you suffer from kidney insufficiency. In that case, your body can’t get rid of aluminum as fast as a healthy body, which leads to a much enhanced accumulation of the metal in your bones and brain. In any other cases, Al in vaccines is most likely (which we know is the scientist’s way of saying “sure as hell!”) safe.
Conclusion
Aluminum is highly problematic as a chronic poison, especially as food contaminant. It is not in vaccines. Anti-vaxxers who claim otherwise confuse acute with chronic toxicity.
Disclaimer: In my blog, I'm stating my honest opinion as a researcher, not less and not more. Sometimes I make errors. Discuss and disagree with me - if you are bringing the better arguments, I might rethink.
Being A SteemStem Member
Great post and I'm glad you addressed aluminum in vaccines. One mildly interesting fact to add is that most aluminum cans are lined with a protective epoxy to prevent aluminum ions from contaminating beer, soda, or any other acidic drinks.
very true, thanks for the ammendment.
There are so many better ways to stay healthy. Just look at vaccines. The fact that there are even payouts should scream red flags. I'm a lawyer as well as comic creator, and I do not buy the settlements are to protect them from bad publicity. If you're product is safe, you go to trial and you defend the damn product. When it comes to products on the marketplace, settlements are because the product is defective. Look at all the payouts here.
• http://www.wakingtimes.com/2017/05/09/whopping-vaccine-injury-payouts-us-fiscal-year-2017-released/
Most of the science done on vaccines is not paid by big pharma, but is independent.
I'm trusting in verifyable open knowledge, and not it vague arguments and interpretations.
Meine Schwester redet schon seit Monaten über die Schädlichkeit von Aluminium. Sie hat sich so darüber aufgeregt, dass sich Aluminiumsalze auch in Deos (Antitranspirants) befinden(da besteht ja auch die Korrelation zu Brustkrebs), dass sie allen aus unserer Familie natürliche Deocremes bestellt hat :D
Fand ich voll interessant. Danke für all diese Überlebenstipps und fürs unterhaltsame myth debunking hehe
Hi, freut mich dass es hilfreich war.
Ich befürchte das ist ein weiterer Mythos, sagt zumindest das national cancer institute:
"Because underarm antiperspirants or deodorants are applied near the breast and contain potentially harmful ingredients, several scientists and others have suggested a possible connection between their use and breast cancer (1, 2). However, no scientific evidence links the use of these products to the development of breast cancer."
Yup, let me rephrase that: Da könnte eine Korrelation zu Brustkrebs bestehen.
Das fand ich auch interessant.
Auch aktuell meinen Forscher, dass einfach mehr Studien dazu benötigt werden.
Aber ich kann mir auch nicht vorstellen, dass Deos mit Al nicht in irgendeiner Form schädlich sind, vA angesichts der Tatsache, dass das ein Produkt ist, das man jeden Tag aufträgt.🤔
NOOOOOOOOOOOOOOOO!
Damn it.
So, to be safe, I should switch to a different way of making coffee now? Or doesn't it really matter in the end, because we are slowly dying by food poisoning anyway?
there are plastic capsules around, but then you get the phtalates. You can use non-aluminium mokka pots, filter or espresso machines, I guess. But I have to add the nespresso-problem is just a theory, I don't think it's a scientifically proven fact.
I'm glad you mentioned solubility.
AKA bentonite clay, which we use as an adsorbent to remove toxins during water treatment, and which has been used as a medicine traditionally ingested by some cultures, with varying effect.
jeder scheiß wird mir angezeigt, da wär mir diese super Artikelreihe fast entwischt. Super freue mich auf mehr
das geht mir auch regelmäßig so. Ab 100 Leuten, denen man folgt, wird's einfach übel, den Überblick zu bewahren, wenn man nicht dauernd im Feed hängt.
Muss ich vielleicht wieder mal aussortieren ;-)
Da musste ich jetzt lachen, weil das habe ich mir auch gedacht. Ich hab' momentan keine Idee, wie eine Liste ausschauen sollte, die ich regelmäßig abarbeiten sollte, um mich auf Steemit an den richtigen Orten zur rechten Zeit herumzutreiben. Jedenfalls bin ich froh, noch vor Mitternacht noch auf diesen Artikel gestoßen zu sein, auf den ich schon gewartet habe!
Interesting and really informative one @sco,
We often use Al utensils but never thought of this. Most of the governing bodies have banned on its use. Thanks for sharing😊
Al utensils are probably fine as long as you don't heat acidic stuff in it, I guess. But that's the one thing you really shouldn't do.
No, they aren't poisoning our kids. Except the ones that developed post infectious polyradiculopathy (ascending paralysis) and died awhile after. Perhaps all the testimonials were frauds, paid off by "anti-vaxxers" but we shun conspiracy theorists! Right!? Maybe the CDC whistleblower was a fraud, just like the CDC told us! In all seriousness though, there are no mulit-generational studies on vaccines, in addition to our limited understanding of the body, this gives cause for skepticism. It serves to reason that ssRNA could be transformed into DNA in the presence of reverse transcriptase resulting in epigenetic mutations. I could argue vaccines are slowing down the evolutionary process as artificial active acquirement of immunity doesn't change the genome, but that of course would lack empathy and you may suggest that without vaccines we wouldn't be here today, citing studies indicating the vaccination savior complex. Again those studies would be contested just as the aforementioned physiological theories would be. In my opinion the truth about vaccines is that we don't know the truth about vaccines. Patronizing "anti-vaxxers" seems a bit ironic on a platform full of people like me who were laughed and scoffed at 3 years ago when talking about "crypto-currency." This is a contrarian platform, vaccines are universally accepted. This is a political issue as well, and I'm a big freedom guy. I like freedom and the freedom it gives me to be free.
Thanks for commenting. However, I'll have to argue with you ;-)
There will never be multi-generation studies on humans on any subject. It's simply impossible. You can do epidemiologic studies, but they have their limits. Doing them in a way that adresses effect over many generations is next to impossible.
Skepticism is good, but hysteria isn't. Especially not when mixed up with with a lack of scientific understanding:
just lol. First of all, this wouldn't be epigenetic. Second, even if this was the case (it isn't), what would it have to do with vaccines? If RT (which is not found in humans, we only have it as a part of another enzyme, the telomerase) would lead to the vertical transfer of genes, every RNA virus would be a huge problem for us, no matter if origined from "natural" infections or vaccines.
That would indeed lack empathy. But besides that, it's an incredible stupid argument, because EVERY medical advancement is changing the evolutionary process. Should we abolish antibiotics, appendix surgeries and hospitals too because of that? I guess we could live a stone-age life again just to strenghen evolution, but why would we want that?
BS. There is vast scientific knowledge about vaccinations. If you refer to the fact that science can never be 100% sure, yeah ok. But that's why we are working with probabilities, and vaccines are 99.99% safe. 0.01% is still a valid opinion, but by no means an equally valid opinion.
So what? The platform doesn't belong to you guys, but to all of us. That's the way it was designed. If anyone else is allowed to spread BS, I am allowed to provide the counter-argument. If your are so freedom-loving, why would you deny me the right the speak freely?
So it wouldn't constitute an epigenetic mutation, it could still happen and have negative systemic effects. I'm not trying to play semantics, but perhaps we should get on the same page. When you say vertical transfer do you mean in the birth process passed on to the next generation? That's not my understanding of epigenetic mutations. Should we abolish antibiotics? Well, no, should we force antibiotics down peoples throats in order to go to school? Many doctors are trying to get away from them anyways (at least with children) for the same evolutionary reason. Comparing vaccines to surgery seems a bit like an apple to an orange. When we break our arm, we don't develop immunity to fractures down the road. Sure, scientists know alot about vaccines, to the extent that our immune system is as simple as taught in med school. I'm suggesting that we have barely scraped the surface on that topic, the only topic we know a decent amount about is anatomy, and even that is unpredictable. What do you say to the parents who lost children to Guillian Barre? All the scientists and all the studies in the world, but what do you say to them? Nothing, you sweep them under the rug and continue to give kickbacks to doctors who keep their patients on a regular vaccine schedule. I'm not trying to deny your right to speak freely, I'm simply pointing out the irony of your tone when scoffing at non-conventional thinkers.
Believing vaccinations don't work and are evil has nothing to do with non-conventional thinking, it's plain denial of scientific facts because you'd rather believe the propaganda of "alternative minds".
You should probably read this. I qoute:
"There is little evidence to support a causal association with most vaccines. The evidence for a causal association is strongest for the swine influenza vaccine that was used in 1976-77."
You will never hear me saying that vaccines are in all cases completely safe. But the vast majority, and especially those that are common today, are. Also, as I pointed out, the use of aluminum is safe.
I have no idea what exact mechanism of mutations you are referring to... a vertical transfer is the transfer of a gene from another organism or virus to the host organism. And it is not more likely to happen from a vaccine than from a natural virus, that's what I was saying.
In the end, it comes to the point that even as vaccines cause harm in very very very few occasions, they are much preferable to the alternative: having all these deseases as epidemies on a regular base.
I'll post this because I wrote it before reading @Sco had said pretty much the same thing...
"post infectious polyradiculopathy ". You mean Gilainne Barre Syndrome? A neuropathy, not a radiculopathy lol... A condition that regularly occurs post C. jejuni and is well-researched post-vaccine and consistently shown to be a result of an underlying jejuni infection?
You don't need a multi-generation study to look at the impacts of something that doesn't affect germline cells.
You don't understand the human body doesn't mean we don't understand the human body.
Reverse transcriptase doesn't just float around transforming random thing lol... and let's say it did... how is that then incorporated INTO our genome? Just a floating piece of dsDNA that slots in?
A "contrarian platform" yeah? Try this new thing called using your brain... being an anti-vaxxer isn't a tough slog mate. It's quite the bandwagon these days. Try your hand at understanding science and medicine, THAT is a tough slog.
Feel free not to vaccinate yourself, but as always we all live in democracies and your freedom is balanced against the dangers it poses to everyone else. If your freedom not to vaccinate poses a risk to herd immunity, then you should probably go live on an island with all the other anti-vaxxers... let's see how long you remain anti-vax after.
"What do you say to the parents who lost children to Guillian Barre? All the scientists and all the studies in the world, but what do you say to them? Nothing, you sweep them under the rug and continue to give kickbacks to doctors who keep their patients on a regular vaccine schedule." -> Honestly this is just a joke... so let's break it down. GBS only rarely occurs in relation to vaccine and no causative link has been established... though there's a strong one with C. jejuni though. And believe it or not, the world is a large place. Not every nation gives kickbacks to doctors who vaccinate, yet every nation vaccinates. I know it can be hard to understand this with such a closed mind, but science exists all over the globe (you do believe it's a globe right?)... and all over the globe scientists aggree vaccines work and are risk-effective.
You seem to have a tentative relationship with reality mate. While you try and put forward logical arguments you stick to your indoctrinated beliefe while @sco mows down your points one by one. It's this clutching at straws that, in the end, always shows anti-vaxxers are never science, reason or logic based. It's just a religion. Like denying climate change or any other nutjob conspiracy.
@tfcoates radiculopathy is a type of neuropathy, im not sure why you needed to clarify what I meant by polyradiculopathy considering i referred to it as Guillian Barre as well. Wasting time with semantics to sound smart and you're wrong. I suppose anti-vax is gaining "steem" maybe that explains the personal attacks in both your comments. Nice to see people question their indoctrinated ideas, probably concerning for people who have been forcing vaccines on others while claiming a moral and intellectual high ground. It's not just me that has respect for the complexity of the body, neuroscientists all over the world will admit they understand very little of the nervous system. My position on vaccines is not indoctrinated, I chose to get vaccinated in the past, I've recently changed my mind and its possible i will again. This argument has elements untouched, i'm not digressing but I should clarify that I'm not entirely against vaccines, just like I'm not entirely against opiods. I don't agree with the CDC schedule and the age at which kids are vaccinated, and I don't see the value in the flu vaccine. Vaccines could very well be something that we look back on 30 yrs from now and go palm to forehead, just like we have with the initial rounds of vaccines. I understand my point on GBS and what you say to the parents is an appeal to emotion, but it's a reality, its not a joke for the families affected. That being said, I appreciate you guys taking the time to come down from your intellectual high-ground and engage with such a troglodyte as myself.
You're right, radiculopathy is a type of neuropathy, but not the type GBS belongs to... we figured that out decades ago.
What explains the person attacks is that a) I'm a dick and b) even only six weeks into actual hospital rotations and I've seen children suffering from vaccine preventable diseases. One bad enough that she'll never fully recover. Stop acting like you're perpetrating a victimless crime by spreading this crap... and c) because all your comments here on steem are visible, including the ones where you call pro-vaxxers 'charlitans'.
Oh and on a site note it's not a Mark Twain quote.
Glad you've finally recognized our moral and intellectual high ground.
Of course families are affected by GBS... if you want to talk about real impacts of vaccines talk about anaphylaxis or serum sickness... both are far more concerning for doctors administering vaccines. That's why they make you stick around 30 minutes after a flu shot. Literally every intervention any health professional has in the arsenal will have side effects, the best we can do is weigh them up against harms and 10/10 times vaccines come out right next to antibiotics as one of our best tools.
I do regret calling them charlatans. There is mudslinging on both sides. This topic ends up being very polarizing, to the extent that both sides are wrong. I do feel its a worthy topic tho, which is why I followed @sco. Im trying to decide where I really stand, im trying to keep an open mind. Im not on steemit for money im here because the content is so much better than facebook (although I have earned about 2$, nice)
I think there should be a balance and I think healthy lifestyles should be emphasized more than vaccines. I don't think either of you have all the answers and I know MD's on both sides of the spectrum. Because of the neurotoxins im not comfortable vaccinating newborns. The risks involved with vaccines seem to be more gnarly than the risks involved with the flu, in america at least. There are a lot of different angles at this debate and I really dont think its as simple as "yes or no"
BTW interesting side note on Samuel Clemens, but Im going to keep giving him credit for it because Im related to him.