Effects of Ozone layer Depletion
The decline in ozone focus in the center layers of the environment – fundamentally in the stratosphere – is amazingly harming to life on earth, and is to a great extent caused by emanations of halogenated hydrocarbons delivered by man, CFCs, HCFCs, halons, carbon tetrachloride and methyl bromide. Thus, such substances are normally alluded to as Substances that Deplete the Ozone Layer (ODS).
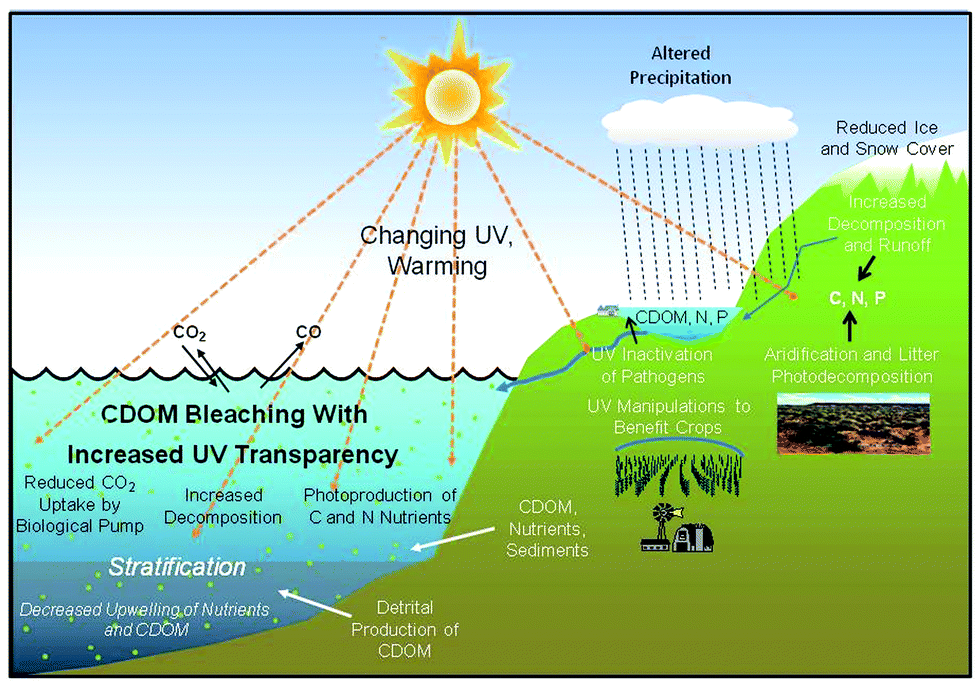
The principal cautioning voice originated from a paper distributed in 1974 by researchers Sh. Rowland and M. Molina of the University of California, who demonstrated that chlorofluorocarbons (CFCs) utilized as a part of refrigeration, cooling and plastic froth producing were in charge of the fast decimation of ozone.
Reason for Ozone Layers Depletion
Hydrochlorofluorocarbons (HCFCs)
Mixes shaped by H, Cl, F and C. They are being utilized as substitutes for CFCs in light of the fact that a significant number of their properties are comparable and are less destructive to ozone by having a shorter half-life and discharging less Cl iotas. Abatements are somewhere in the range of 0.01 and 0.1. Be that as it may, as they stay hurtful to the ozone layer, they are viewed as just an impermanent arrangement and their utilization has been restricted in created nations since the year 1930.
The ODS
There are additionally other synthetic substances that are for the most part gathered as Ozone Depleting Substances (ODS). Illustrations are methyl bromide use in pesticides, methyl chloroform utilized on making modern solvents, and halons utilized as a part of flame quenchers. Much the same as the chlorofluorocarbons (CFCs), these substances likewise synthetically respond with the ozone which begins a compound cycle that separate the great ozone.
Methyl bromide (CH3Br)
It is an exceptionally powerful pesticide that is utilized to treat soils and in numerous harvests. Given its substance in Br harms the ozone layer and has an ozone exhaustion capability of 0.6. In numerous nations dates have been set around 2000, from which it will be restricted.
Chemicals
Different synthetic concoctions that normally give comparable responses the great ozone incorporate Clx, Hox and Noy which have a place with the Chlorine, Hydrogen and Nitrogen families separately.
Thats why world temperature is increasing
Yeah thats are the reasons
Thanks for sharing this ifo
your most welcome
Congratulations! This post has been upvoted from the communal account, @minnowsupport, by afzaal4 from the Minnow Support Project. It's a witness project run by aggroed, ausbitbank, teamsteem, theprophet0, someguy123, neoxian, followbtcnews, and netuoso. The goal is to help Steemit grow by supporting Minnows. Please find us at the Peace, Abundance, and Liberty Network (PALnet) Discord Channel. It's a completely public and open space to all members of the Steemit community who voluntarily choose to be there.
If you would like to delegate to the Minnow Support Project you can do so by clicking on the following links: 50SP, 100SP, 250SP, 500SP, 1000SP, 5000SP.
Be sure to leave at least 50SP undelegated on your account.
Sneaky Ninja Attack! You have just been defended with a 3.23% upvote!
I was summoned by @afzaal4. I have done their bidding and now I will vanish...
woosh
A portion of the proceeds from your bid was used in support of youarehope and tarc.
Abuse Policy
Rules
How to use Sneaky Ninja
How it works
Victim of grumpycat?
You got a 7.00% upvote from @dailyupvotes courtesy of @afzaal4!
Great post!
Thanks for tasting the eden!
This post has received a 1.88 % upvote from @booster thanks to: @afzaal4.
This post has received a 6.96 % upvote from @boomerang.