Is depression an exaggeration of body's healing mechanisms - Taking a dive into depression and inflammation.
As time passes, more and more evidence is accumulating that suggests that depression is not just a mental state. It has associated physiology to it that has gone haywire. There is associated pathology of neurons inside a depressed brain and there is a contribution from the inflammation in the body. But have you ever wondered how these two are connected? Let's try to figure out.

Adapted from Image by EvgeniT | Pixabay.
Previously on depression and inflammation
Last year I shared a post titled - This is what depression is made of! - The biological underpinnings of depression. In that post I discussed the various pathological mechanisms linked to depression. The mechanisms included changes in dendritic spines (the input terminals on the neurons) and the inflammatory status of the body. I discussed how the microglia (the immune cells of the brain) are chronically activated in depression and they secrete inflammatory cytokines such as TNF alpha, IL6, interferons etc. I also talked about the evidence regarding TLRs on the cell surface of immune cells getting activated before secretion of these cytokines. However, if you blocked the TLR signalling you can rescue depression-like symptoms in mice. I also told you about Rag1 null mice (the mice which lack T cells). We saw how T cells are important for the resolution of inflammation in the body and in lack of T cells, the Rag1 null mice have worse symptoms of depression.
So far so good. We have good evidence that inflammation and depression are somehow related. However, what we hardly know about is how and why in the freaking world they are related? This is what we are going to discuss today. The mechanistic insight.
Why would inflammation cause depression.
The origins of depression.

Adapted from Bandaid by Clker-Free-Vector-Images and Brain by Stephanie_Curry | Pixabay.
In many cases when a disease is not caused by infection, it's probably an altered regulation of something that is bound to happen normally. For instance, one can look at solid tumours as overhealing wounds. In a similar sense, symptoms that fall under the umbrella of depression - consistent sadness, social withdrawal, constant tiredness, lack in activities that once brought pleasure to you - might have been an adaptive strategy to avoid adverse life situation (Hagen et al., 2011). And perhaps when this kind of short term adaptive behaviour is chronically activated, due to some malfunction in the body we see a long term disease. Some people have gone to the length of suggesting that depression evolved as a strategy to give you time to analyze the problem (see this article by Andrews and Thomsom).
But of course, it is hard to see this short term adaptive strategy as something "evolutionary". How does something that decreases your reproductive fitness even get selected? Think about it this way - while you wallow in your sadness for years, not having any interest in sex, the other members of your species would simply outnumber you (Sharpley and Bitsika, 2010).
Well of course, unless there is something else going on. For instance, inflammation. Think of it this way - you are a deer or zebra in the jungle being chased by a predator. After that long chase or tough fight, your body may want to turn on the repair mechanisms that would heal the wounds you might have endured. Or infections you might have caught. All these repair and healing mechanisms and fighting microbes require an immense amount of energy by the immune system. Hence, one likelihood is that after that fight or flight response, your immune system bias the energy use towards the healing process; rather than looking for mates, enjoying meals or even case of humans hanging out with friends. This might explain why these symptoms of depression look adaptive in the short term.
However, now imagine a situation where these inflammatory processes are activated chronically. For instance, by chronic stress (see previous blog). Well, yeah humans and animals can turn on low-grade systemic inflammation just by overthinking shit (Miller et al., 2019, Liu et al., 2018). Or think of a scenario where this inflammation is chronically activated by the gut microbiome, obesity, cardiovascular disease or some autoimmune disorder. All of which, by the way, can be correlated with depression via inflammation (Pryce et al., 2017, Tyrrell et al., 2019, Halaris, 2013, Peirce and Alvina, 2019). The question that we need to ask is that is there a mechanism by which inflammation, say inflammatory cytokines, irrespective of why they were there can explain symptoms of depression?
Cutting down the spending on fun!
Let's take the loss of interest in pleasurable activities. That sheer lack of motivation to get out of bed and do something that makes you feel happy? Or would it even make you feel happy anymore? How on earth can immune cells and molecules they secrete possibly change how you use to perceive pleasure?
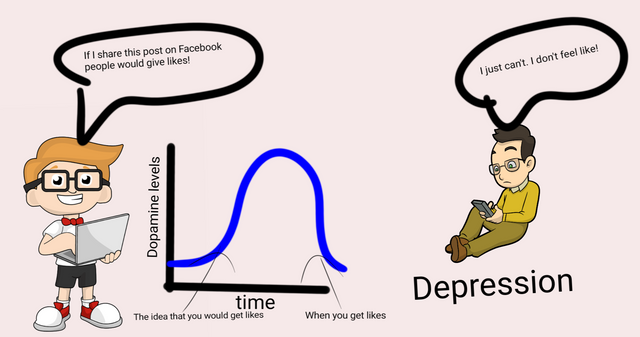
Illustrated by @scienceblocks using the following images -
Image of guy with laptop | CC0
The depressed guy on mobile by Free clip art | CC BY-SA 4.0
Well, I am sure you must have heard about Dopamine at some point in your life. Dopamine is a neurotransmitter in the brain. In part of your brain, there is an entire network of dopaminergic neurons (neurons that secrete and communicate via dopamine), which is involved in anticipation of a reward. What this means is when you get a sensory cue - say a tone that requires you to say raise your hand. Your brain is going to process the outcomes of it. Like, what would I get if I raised my hand? Will there be a pat on the back, chocolate, a kiss, an orgasm following this? Is it worth raising my hand? Well, that kind of calculation. If dopaminergic neurons fire, it means you are anticipating the reward and you are likely to raise your hand (see this video by Robert Sapolsky).
No wonder, a chronic drop in dopamine levels or perturbation of the dopamine pathway is associated with depression (Belujon and Grace, 2017). Especially, when it comes to explaining the symptom of anhedonia (lack of interest in pleasurable activities), in patients. However, can chronic low-grade inflammation in the body explain the decrease in dopamine activity in the brain?
Turns out it can. In the previous blog, I talked about how INFalpha (an inflammatory cytokine) therapy, given to patients with viral infection and cancer, can also cause depressive mood disorder. Turns out INFalpha causes a decrease in dopamine activity in the brain. You can also administer INFalpha to non-human primates or mice and induce depression-like symptoms in them. INFalpha administration also turns on production of INFalpha, IL1, IL6 and other inflammatory cytokines in the brain. It has been shown in different experiments that these cytokines can reduce the availability of a molecule called BH4. This molecule is required by the enzyme in neurons that synthesize dopamine. Moreover, these inflammatory cytokines have also been shown to downregulate the protein VMAT2. This protein is required for packaging of dopamine for release from neuron synapse. Moreover, inflammatory cytokines have also been shown to affect the expression of proteins involved in dopamine transport and its reuptake by the neurons. Even though the exact pathways for all these mechanisms have not been completely elucidated, I would say there is good evidence for inflammatory cytokines being able to disrupt the dopamine signalling in these neurons (Felger and Miller, 2012). Perhaps, we need to pursue this further.
Moreover, that is not where the story ends. Accumulation of dopamine inside neurons creates a stress response. This can cause these cells to produce reactive oxygen species, which is known to induce inflammation (Felger and Miller, 2012). This would set in a vicious cycle. Also, this would mean that it doesn't matter where the depression initially starts in. If it was an external stimulus from inflammation or whether due to mental stress you accumulated more dopamine inside of these cells and set in the inflammation in the brain. You might be caught up in that physiological cycle now. Of course, you can genetically or epigenetically be more predisposed to inflammation in the body or there was an issue with genes that clears up dopamine. Which would explain why some people are more or less resilient in developing depression. Also, why epigenetic modification of stress response and immune profile by early childhood or prenatal life stressors can make you more or less likely to develop depression (see this and this).
No strength for physical activity - the chronic fatigue
It is also to be noted that dopamine in basal ganglia in the brain is also involved in controlling motor activity. Hence, interruption the dopamine signalling will not only cause unwillingness for you to raise the hand, but you will raise your hand quite slowly. You will feel the fatigue all over your muscles which would fuel you unwillingness even further. And that's how the link between inflammation and dopamine signalling would also explain another symptom of depression - the chronic tiredness (Fleger et al., 2017, Lee and Guiliani, 2019). Also, don't forget that inflammatory cytokines also affect metabolism in rest of your body (see this blog). So, on one hand, they affect the psychomotor pathway, on the other hand, they may also reduce the energy availability of other cells including your muscles.
Is inflammation trying to reduce your energy expenditure?
This brings us back to our initial question. Is depression an exaggeration of pathways that are supposed to help you heal? Is that why inflammatory cytokines turn off your motivation by suppressing dopaminergic activity? Is that why they suck the energy out of the motor activity so you just lie down and stare at a wall till you heal? Treadway et al., 2019 suggest that that is perhaps what is going on. They also proposed a computational framework considering immunometabolic state as an input variable in the decision-making process to put effort into something. What they suggest is that the dopaminergic system takes input from the immune system and changes the perception of effort. Instead of thinking will I or won't I make an effort to do something, you will think can I or can't I make an effort to do something. And then, in depressive disorder you will mostly end up feeling - I don't think I can! And there they are, the inflammatory cytokines changing the way you think about whether or not you have the energy to do something. They changing your perception of effort and reward altogether.
Altering the emotional states.
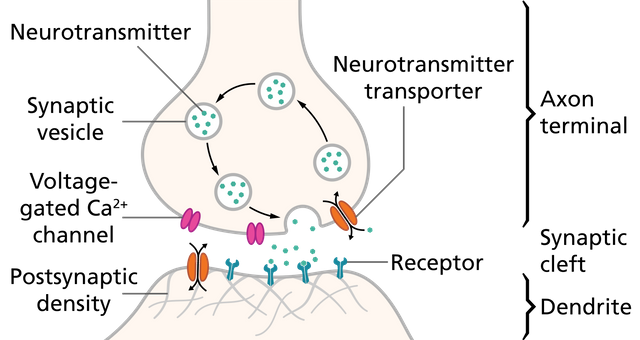
Image by Thomas Splettstoesser | CC BY-SA 4.0
Now, to say dopamine would explain entire depression would be wrong. Of course, there are other pathways at play here. For instance the serotonin pathway. The exact role of serotonin in depression is not very clear, but we know drugs used as antidepressants are selective serotonin reuptake inhibitors (SSRIs) - such as Prozac. That means that they increase the levels of serotonin in synapses of neurons that communicate via this neurotransmitter. These drugs work slowly over time, but they seem to work in many cases. The first question should be how? From computational studies, it appears that serotonin may be involved in inhibition of negative thoughts. Evidence also hints that SSRIs may act by improving the emotional states on a short term basis (perhaps by blocking negative thoughts). Which may explain why they take so long to act (Cowen and Browning, 2015).
What inflammation does to serotonin pathway is that it leads to conversion of an amino acid tryptophan to kynurenine. Tryptophan is required by serotonergic neurons to synthesize serotonin. One of the assumptions in the field is hence that inflammatory cytokines activate the enzyme (IDO) that causes this conversion. The reduced tryptophan then causes a reduction in serotonin synthesis. Though more solid evidence is required for this. However inflammatory cytokines such as IL6 and TNFalpha also causes an increase in gene expression of serotonin reuptake transporter. If this transporter is increased then it would cause a decrease in the availability of serotonin in neuron synapses (Miller et al., 2009).
But, how can turning on negative thoughts by interfering with serotonin, fit into the picture of energy expenditure hypothesis for depression? I mean sure negative thoughts may demotivate you from doing a lot of things. Say you wanted to post your picture on facebook, but then you think - you are ugly, nobody would like you, they would laugh at you. You know those kinds of thoughts. But if you are thinking negatively you would be stressed and maybe even anxious. Those tensed muscles, stressed heart and all would consume some good energy? No? I think someone needs to work on this aspect. However, one possibility is like what happens with the food we eat during fever or context we encounter during bad health or bad break up. We develop a distaste for that food and fear for that context. We tend to avoid it in future. And when you chronically activate the negative thoughts, chances are that you will develop anxiety or generalized fear which is also found as a co-morbidity in depressive disorder (Hirschfeld, 2001).
Is learning expensive or difficulty learning new things is a price we pay?
Moreover, can Inflammatory cytokines explain cognitive distortions such as difficulty in learning and recalling new information during the depression (Kizilbash et al., 2002). Seems like it can. Hippocampus is the brain region which helps us in learning new stuff and even in memory recall (along with some other cognitive skills). We know from neuroimaging studies that there is a significant reduction in hippocampal structures in depression (Roddy et al., 2019). Inflammatory cytokines at low levels seem to have beneficial effects on neurons in the hippocampus. But prolonged exposure these high levels these cytokines and glutamate imbalance (which can also be caused by kynurenine), can hurt the neurons in the hippocampus (Miller et al., 2009). So what is going on here? Are cognitive abilities, such as learning, down in depression because of the exaggerated healing process in the body? Is this also a way for the immune system to snatch a big chunk of energy for itself? Or if not a grand plan of inflammation, are changes observed in hippocampus just a detrimental effect of the disease? It is something worth exploring for sure.
Summary
In a nutshell, what we tried to understand today is a mechanism by which chronic low-grade inflammation in the body can explain the symptoms of depression. We picked up an evolutionary lens to see why when inflammation in the body goes up, the mood and cognitive abilities may suffer.
Inflammation is a sign that the body may be trying to fight infection or heal wounds. Probably, the healing mechanisms evolved to skew the energy use in their favour. So while fight and repair go on you don't invest energy in other expensive tasks - such as pleasurable activities, physical activities and learning new information. At the same time, some energy might have been spared for overthinking negative thoughts, just you don't eat the same food that you ate in sickness again or you won't go in predator territory again. Which may also explain why on one hand the learning center hippocampus suffer in depression but amygdala may be learning new fear memories.
It's interesting that some inflammatory cytokines alone are sufficient to turn on these depressive symptoms. In this post, we discussed some of those mechanisms and cited papers where you can get more information on them. There are doubts that depression is not just in your head, its also in your immune system. What we saw is a likely exaggeration of normal processes that could be useful in normal healing following - stress, sickness and wounds manifesting as a disease, when chronically activated.
About steemstem
But, before I go I would like to mention about the steemstem platform. Well, if you love reading and writing interesting science articles @steemstem is a community on steem that support authors and content creators in the STEM field. If you wish to support steemstem do see the links below.
You can vote for steemstem witness here -Quick link for voting for the SteemSTEM Witness(@stem.witness)
Quick delegation links for @steemstem
50SP | 100SP | 500SP | 1000SP | 5000SP | 10000SP
Delegating to @steemstem gives an ROI of 65% of the curation rewards.
Also, if you have any questions regarding steemstem, do join the steemstem discord server.
You can DM me on discord, I have the same handle - @scienceblocks. Also if you are not a steem user, and reading this blog inspired you to start your science blog, find me on discord and let me know about you. I can try and help you navigate your way through steem.

References
This is what depression is made of! - The biological underpinnings of depression
The evil plan of cancer to hijack wound healing machinery.
Andrews and Thompson, 2009. Depression's Evolutionary Roots
Miller et al., 2019. Chronic stress induces persistent low-grade inflammation
Tyrrell et al., 2018. Using genetics to understand the causal influence of higher BMI on depression
Peirce and Alvina, 2019. The role of inflammation and the gut microbiome in depression and anxiety
Type 2 Diabetes - from the eyes of immune cells.
Treadway et al., 2019. Can’t or Won’t? Immunometabolic Constraints on Dopaminergic Drive
Kizilbash et al., 2002. The effects of depression and anxiety on memory performance
Thanks for this write-up! This is actually a long-term follow-up: one year between each episode! Your readers may need to be patient... ;) I actually vaguely remember having read something about depression connected to inflammations, but I was not remembering well. I am happy with the summary in the post. This seems to make sense (actually the entire post was quite comprehensive, as said by @abigail-dantes), at least for the non-specialist who I am.
Instead of asking questions (as I think I have mostly followed everything), there are two items in the post that made me laughing and that I want to emphasize here.
This sounds weird to my ears: “please don’t be depressed or you won’t transmit your genes”
On different grounds, I have learned that anhedonia was a word today… :D
Lol, I know I have sitting on this article for quite sometime. It happens a lot of times that I would start writing something then wound either lose interest or would think an it's not quite there yet. I would then save it in my email and start thinking about something else.
Haha, not that I wanted to say please don't be depressed or you won't pass on your genes. But it does sound funny when you put it that way. 😁
Also, thanks a lot for this feedback. It is always helpful to know if I was able to deliver the message to everyone. :)
I think I am a good guinea's pig for testing how the post is comprehensive. There is of course a more technical part, but it was fine.
Fascinating @scienceblocks! There are indeed many pathways to depression. I was particularly interested in the disturbance of dopamine synthesis due to inflammatory cytokines. Mostly because it considers and clarifies the kick start of depressive symptoms resulting from both internal and external stimulus (e.g. stress, inflammation).
The energy expenditure hypothesis explained by means of negative thoughts prompted by interfering with serotonin reminded of the principles of evaluative conditioning: the change of feeling towards a stimulus after pairing it with another that carries a stronger or opposite response. It is quite clear!
A truly comprehensive means to address depression and its signs :D
Very enlightening indeed! Thank you @scienceblocks
Yeah way too many. It would take an entire book if we were to discuss all that is involved in depression. How they are linked to each other and also to external factors. Say glutamate pathway for instance. Then whole hypothalamus and insula involvement in regulating appetite and changes in tastes during depression. There is so much going on and we have so little time to understand it all.
Hi @scienceblocks: a brilliant piece. Material was clear and relatable. Nice balance between science and accessibility.
I'm not at all surprised by the relationship you draw between chronic inflammation and depression. One disease with which I have some familiarity is SLE. The SLE story is inflammation, as you know--everywhere/or anywhere. An acknowledged symptom of SLE is depression/anxiety. Studies on SLE patients back up your thesis... a demonstrated connection between chronic inflammation and depression.
Your article really laid out a map for exactly how the inflammation interferes with signalling.
Another great piece from the inimitable #scienceblocks:)
Fascinating @scienceblocks! There are indeed many pathways to depression. I was particularly interested in the disturbance of dopamine synthesis due to inflammatory cytokines. Mostly because it considers and clarifies the kick start of depressive symptoms resulting from both internal and external stimulus (e.g. stress, inflammation).
The energy expenditure hypothesis explained by means of negative thoughts prompted by interfering with serotonin reminded of the principles of evaluative conditioning: the change of feeling towards a stimulus after pairing it with another that carries a stronger or opposite response. It is quite clear!
A truly comprehensive means to address depression and its signs :D
Very enlightening indeed! Thank you @scienceblocks
I recently read about depression contributing to pain but I have not yet heard about pain / inflammation contributing to depression.
For clarity, I am referring to clinical depression in the former case, not what we in our colloquial may refer to as being depressed or sad. (Although, my pain can prompt such feelings)
I'm going to review this more closely but based on the premise as I understand, I need to make sure to stay on top of taking my anti-inflammatory medications and / or more holistic remedies to assist my body in healing.
Caveat: I understand there may be issues with NSAIDs so would highly recommend people looking into remedies such as Tumeric.
I think it's more likely that they both fuel each other. I mentioned one such possibility in the article such as inflammation-Dopamine accumulation-ROS generation-more inflammation pathway. Similar axis could also exist for cell damage caused by depression. Then there can be behaviour alteration, where poor lifestyle habits may fuel further inflammation.
I have heard of reports on depression altering pain perception. However, there are also reports on pain-sensing neurons being able to turn on the inflammatory response (I did a small post on this). Though at the moment I am not aware of the nociceptor-inflammation-depression axis. Something that should be looked into, however.
Ah! Turmeric. I remember as a child, having to drink a glass full of turmeric dissolved in hot milk. I would get that every time I would get wounded while playing. Quite recently someone gave it to me the last time I caught a common cold. I was fine the next morning, though hard to say if it was meds or if it was the yellow latte. I find this suggestion of turmeric very interesting and would love to explore the possibility.
Thanks for for your insightful comment, I have a lot more questions in mind now. If you happen to find something interesting during your review, please share. :)
Great post!
Resteemed for more visuability. :)
Amazing content. Thanks for sharing your knowledge about this topic. It made me remember some of my experiences when I got depress and my routine to reverse my lack of motivation.
Posted using Partiko Android
This post has been further promoted on Facebook, Instagram, Reddit and Twitter by the SteemSTEM team!
Fascinating stuff, there is increasing evidence that inflammation is linked to a whole host of diseases such as heart disease. Really appreciate the comprehensive approach, keep up the great work.
Hey, this is a great article.
This is pretty much the explanation for Depression in Traditional Chinese Medicine.
Whether you agree or don’t agree on the methods of treatment (herbal medicine and acupuncture), the understanding of how the body works can shed light on stuff like this.
The problem most (Western) Medical scientists have is the metaphorical and flowery language of ancient China.
It is quite literally ‘lost in translation’....
When I was working clinically, I always worked with ‘Depression clients’ in this way, usually by finding dietary/lifestyle triggers for inflammation. I found giving them medicines or ‘treating’ them wasn’t anywhere near as effective as simply removing what triggered inflammatory responses.
Thanks 😊🙏🏽☯️
Posted using Partiko iOS
This is interesting. I don't know much about traditional Chinese medicine and how it explained about inflammation and depression. Though your comment makes me curious. I would try to explore this a bit. I think you are on right path with your clients. Though, I am curious to know about your depression clients. How did the dietary lifestyle changes worked for the. If you ever quantified their progress. What percentage of them showed improvement. If you want to do, share with us their stories. It would be nice to read about their progress.
I’ve retired from the profession now, and I don’t have any old files or anything like that to refer to... sorry. Otherwise, it would’ve been great, provided they consented to their info being shared of course...