Childhood stressors and the beginning of pathogenesis chain reaction(Part 1 - Glimpse of the Hackathon, inside the womb)
Maybe, what happens in Vegas, stays in Vegas. But, that is because you are an adult. However, what happens in womb and childhood, goes with you everywhere. And this is what this post is going to explore. Why is childhood so important in terms of our entrire life. Why bad experience during childhood pushes us to both mental and physical illness, in our adult life.
Image by DanEvans | CC0
In the previous parts, about how psychology affects psysiology, I mainly focused on development of type 2 diabetes. In part 1, I told you about link between childhood stress and metabolic disorders. In part 2, we explored the mechanisms by which psychological stress alters physiology. However, I did not focus on stress during early life. Well, because I like to keep the articles short, and also because this deserves an article of its own; and you will see why.
To understand why events in our early life, are so important in terms in shaping the rest of it; you need to understand, what is going on in our body at that time. Even if you are born in this unpredictable world, with genes identical to your twin sibling, your life can still take different routes. The routes can be extremely different , in terms of mental and physical health, based on stressors in your environment.
It all starts in the womb.
Of gene expression and generations of Hongerwinter
Image by OpenClipart-Vectors. |CC0
In 1944, a famine took over German occupied Netherlands. They called it Hongerwinter, or hunger winter if you translate it to english. The dutch hunger winter became of particular interest to biologists. What have biologist got to do with dutch famine, you ask?
The pregnant women, who were in mid-pregnany, gave birth to children who were going to change how we thought about meternal environment. The kids born were of more or less normal weight. However, 1976 they found that these kids were more likely to be obese and have metabolic disorders(Ravelli et al., 1976). In 2008, Heijmans et al., proposed that these disorders were because starvation during this period of pregnancy changed how different genes are expressed in body.
Doodles by @scienceblocks
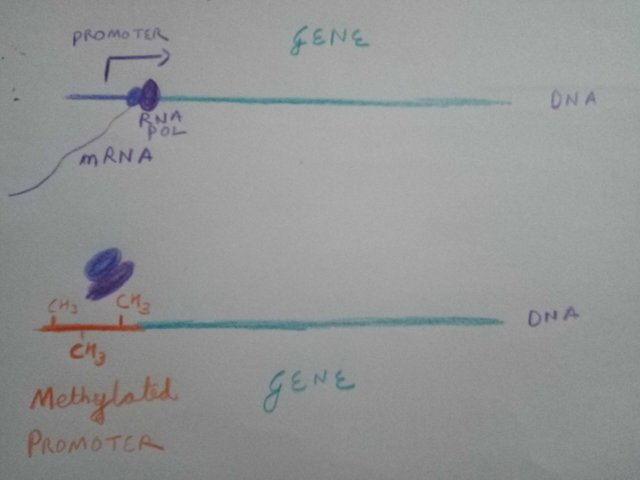
So, it's not just about having the genes, but also about how those genes are expressed. The expression changes can occur by adding or removing methylation from the promoter region of genes or by making chemical modification to histone proteins that package the DNA (Watch the Epigenetics video below). So, what if you add a metyl group to the promoter region? Well, the gene will be expressed at lower rate or not at all. If you remove it, you can increase the rate of gene expression. Well some people call epigenome the software of genomic hardware of the cells. I like to think of it more like ROM of the genomic computer. (Also, note that there are other extra genetic regulation important in what I am talking about. Such as microRNA regulation of mRNA and mRNA editing. However, for the sake of simplicity I will leave it for exploration of those interested in depth).
One such gene, found to be demethylated in case of Dutch hunger winter born kids, was Insulin like growth factor 2(IGF2). And, overexpression of IGF2 has been shown to cause metabolic diseases and even dysfunction of cells that produce insulin(Casellas et al., 2015). And these scars in the genes were found when these people were 60 years old after being exposed to Dutch famine. Right, 6 decades after Hongerwinter.
You can also take mice, starve them during pregnancy and replicate the Dutch hunger winter's aftermath in the lab(Jones and Friedman, 1982). Moreover, I need you to pay attention to the fact that these epigenetic changes are not just about one generation. But these changes persists and can be inherited even by grandchildren in both rats( Radford et al., 2014), and in grandchildren of Dutch hunger winter's pregnant women(Veenendal et al., 2013).
Nonetheless, that is just about the starvation stress. However, how does other phychological stressors in pregnant mothers afffect or program our genes?
Psychological stress and the future of the Unborn.
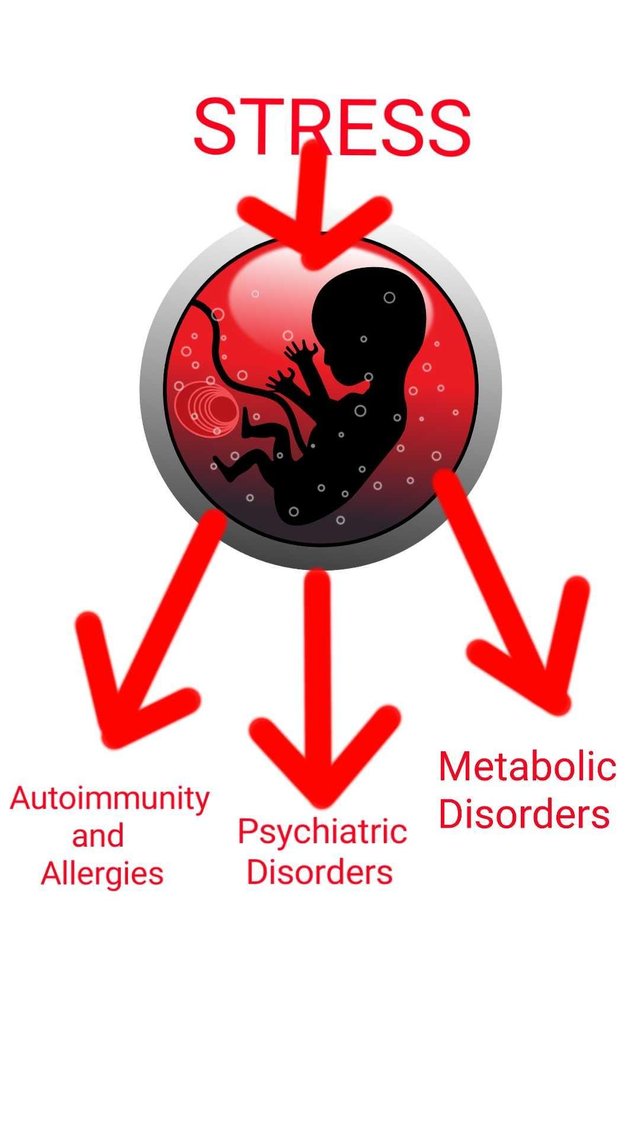
It has been shown that psychological stress faced by pregnant woman is reflected in adulthood of child. The stress hormone cortisol produced by stressed women, do not only affect them but also the child in her womb. The neurodevelopmental and psychological disorders are first hit(Talge et al., 2007).But that is not where the story ends. Obesity and metabolic disorders have also been linked to stress faced during pregnancy(Entringer et al., 2012). Moreover, many autoimmune disorders including lupus can be correlated with inflammatory cytokine pattern observed in prenatally stressed individuals(Entringer et al., 2008). Allergic diseases like asthma and even food allergies are more common in these individuals.
The fetal programing of stress response.
@scienceblocks
So, what is common between mental disorders, allergic diseases, obesity, type 2 diabetes and autoimmune disorders? The obvious one is altered immune response. But what alters it and how?
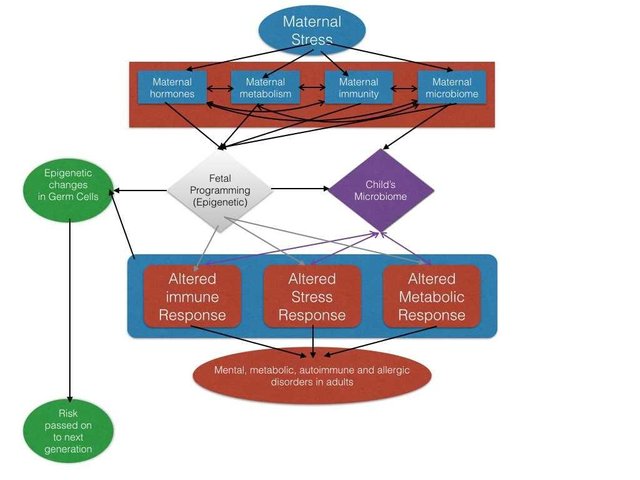
The story goes back to epigenetics again. From replicating the human observation in animal models, one thing looks pretty straight forward - while in the womb, the cells of fetus are already trying to makes sense of how the external world is like. For instance for hongerwinter mothers their fetus thought that there is less food in the outside world. So, they turned on or accelerated the expression of genes, that may help in better food storage.
In fact, the epigenetic regulation of the entire stress response pathway is altered (Monk et al., 2016). Being exposed to stress in mother's womb seem to dull down the response to future stress hormones for life. Especially, the late response, which comes after early fight and flight mode. It looks like your genes are thinking there is always going to war in this world. For instance, the body's response to glucocorticoids, takes the major hit(Oberlander et al., 2008).
Programming the Immune response.
Adapted from an Image by Ricinator | CC0.
Modified by @scienceblocks
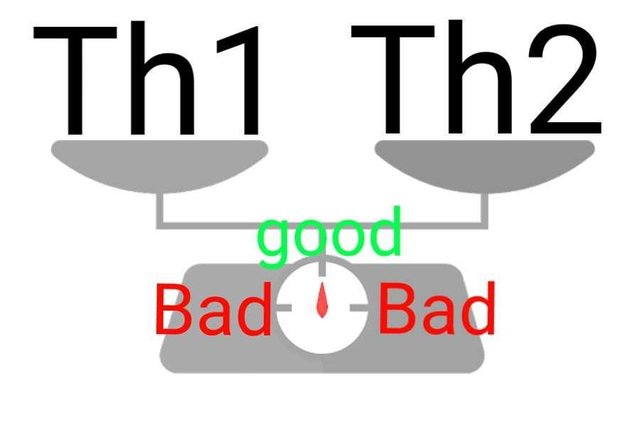
We already saw, how prenatal stress can alter the inflammatory cytokine profile of adults(Entringer et all., 2008). Well, it does look like that exposure to prenatal stress, epigenetically programs the immune system to respond a certain way. For instance, a lot of inflammatory cytokine genes are under the regulation of a protein called NfKb. But if you methylate some of these genes, while demethylate other you can shape your immune system to respond in certain kind of way. When it comes to The T helper cells for example, they can mount two kind of immune responses - Th1 type and Th2 type. The imabalance between these, seems to associated with many stress related pathologies. And Th2 type is what prenatal stress programs the immune system for(Cao-Lei et al., 2016).
Moreover, remember the chronic low-grade inflammation that I talked about in my previous blogs, and it's relation to metabolic disorders? Well it looks likes prenatal stress is also linked with sysyemic inflammation in adults(Slopen et al.,2015).
So not only you have a bad stress response for your adulthood, you have a pathological immune system, waiting to deploy an immune response more in direction of mental, metabolic, allergic and autoimmune disorders.
Programming the energy homeostasis.
Remember, that in part 2 of how psychology affects physiology, I told you about stress alters, how body percieves energy requirements. Like zebra being chased by predator, should better have more energy to run. So what if the mother's body is telling you that your energy requirements are going to be pretty high, in this crazy world out here. Well, you better be prepared. Epigenetic patterns, on genes involved in energy homeostasis, are also different in those people, whose mothers were stressed or depressed during pregnancy(Thorsell and Nätt, 2016).
Deciding the microbiome of adults during their fetal life.
In my blog, about type 2 diabetes and microbiome, I told you that composition of your microbiome is function of your genes. I also told you that microbiome can affect the gene expression in your body via epigenetics. However, does fetal epigenetic programming have any affect on microbiome composition of the individual?
Since, we already saw the evidence of altered immune and metabolic programming in fetus of stressed mothers, it should not be very surprising if it does. In fact, nutritional stress of mothers has been shown to do this. For instance zinc in diet of pregnant rats can also alter the methylation of cells in the fetal gut itself. This makes the gut environment more anti-inflammatory, which might select a different kind of microbiome altogether(Indrio et al., 2017).
However, epigentics is not the only way to alter the microbiome. Remember, in the blog in mentioned above, I told you about how microbiome of cesarean and normal babies is different. I also told you that the skin microbiota found in dominance in gut of C-section babies contribute to diabetes and obesity. Well turns out it also contributes to mental illness as well. But we talk about that some other time. However, what if you changed the the vaginal microbiome of the mother itself? Then even the babies born via normal delivery, would also get the altered the microbiome, for starting their life.
Turns, out the mother's vaginal microbiome is altered if they are stressed or depressed. And, this microbiome can explain a lot of long term adulthood maladies. Infact, you can just simply transfer the altered vaginal microbiome of stressed female rats to healthy rats. What happens? Looks like it is sufficient to induce the effects of chronic stress in adulthood(Jašarević et al., 2018).
@scienceblocks
So, we can see the there is plenty evidence out there linking stress during pregnancy to misery of these unborn babies when they grow old. These unborn babies are just preparing for the world out there. The hunger winter babies expected that there would be less food in the world. But that was a transient false alarm. They had gene profile activated to fight starvation and store more food in body. But there was enough food in the world without famine, they later ended up growing in.
Overall, I think it would be a nice idea to take care of your partner needs when you are pregnant. You being around making sure stressors don't disturb her much would be an awesome idea. Not just for you, but for a healthy world. And not just for your child, but also for your grandchild. Moreover, healthcare professionals dealing with single mothers or prospective mothers they think show signs of depression and anxiety should consider the health of future citizens. Considering counseling would not be a bad idea in such cases.
Anyway,that was mostly about how the body can be primed for diseases by stress, even before the birth. In part two let's talk about how childhood maltreatment causes both mental and physical diseases in adults.
PS: I wonder if you still like to separate mental diseases from physical. To my eyes they are all physical disorders. But because most people I know, draw a distinction between the two, I talk of them as two different things. I hope my articles may have atleast made you think otherwise.



I knew it! My research has pointed in this same direction for quite a while. It's wonderful to have confirmation of such. Thank you so much for this article, it is vindicating in the extreme. <3
This may be one of the best articles I've read on Steemit, but it makes me profoundly sad. I think of how readily we allow children to live in poverty. I think of all the stress that a life of poverty entails. It's a lifetime, a inter-generational burden we place on these children.
I wish people understood this better. Thanks for putting this out there.
Resteemed.
Thankyou. I wish so too. Hope the awareness about this reaches more public platforms. Thanks for sharing this further.
This post Is well researched with great references throughout. I’d only add that epigenetic modifications like methyl groups are difficult to detect with many sequencing platforms which is why a lot of this information is only available recently.
Well first of thanks a lot. Your comment means a lot and it's quite motivating. And, yeah I totally agree about difficulties of methylene seqencing. Even when I started my PhD we had hard time to find a good facility for it.
@scienceblocks , if I'll get pregy I will remember this post😊
Thankyou for such a sweet comment.
This post has been voted on by the steemstem curation team and voting trail.
There is more to SteemSTEM than just writing posts, check here for some more tips on being a community member. You can also join our discord here to get to know the rest of the community!
Both mental and physical diseases bring body even life disorder. If not for scientific purpose, there will no reason for differentiate between them.
Precisely. I think what I am also trying to bring forward with my posts that "it's not just in the mind". You know. I appreciate you putting it in perfect words. Thankyou.
Talking about quality post, this is one!
Well researched and written.
Hi @scienceblocks!
Your post was upvoted by utopian.io in cooperation with steemstem - supporting knowledge, innovation and technological advancement on the Steem Blockchain.
Contribute to Open Source with utopian.io
Learn how to contribute on our website and join the new open source economy.
Want to chat? Join the Utopian Community on Discord https://discord.gg/h52nFrV
Resteemed your article. This article was resteemed because you are part of the New Steemians project. You can learn more about it here: https://steemit.com/introduceyourself/@gaman/new-steemians-project-launch