Engineering Human B Cells With CRISPR
Today we will be discussing a recent publication in Nature: Scientific Reports titled "Engineering of Primary Human B cells with CRISPR/Cas9 Targeted Nuclease." I hope you will enjoy this summary, as we go a little bit through the background and material covered in this publication.
Human B Lymphocyte [Flickr.com]
B Cells and The Human Adaptive Immune System
This article focuses on the B Cell, aka B Lymphocyte, which is a cell involved in our adaptive immune response. When we say adaptive immune response we are referring to the type of response which is custom tailored (B cells secrete antibodies) to specifically target the invader. This is to contrast to our innate immune system which is not tailored to the specific invader but rather utilizes macrophages to attack any non-self material.
In a bit of un-intentional serendipity this article focuses on the application of CRISPR (the bacterial adaptive immune system) on those B Cells. However when people talk about CRISPR they are not really referring to CRISPR, but rather talking about the enzyme (RNA Guided Nuclease) Cas9 which is actually one piece of one variety of CRISPR.
What Is CRISPR Anyway: CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats, and actually refers to a bacterial genomic region where the organism stores pieces of foreign genomes (from viruses, or exogenous plasmids), the foreign DNA is stored in between repeating palindromic sequences of DNA, hence the name. This stored foreign DNA gets transcribed into RNA by the cell, and that RNA is used as a guide to direct the nuclease portion of the system to attack and cut up the foreign DNA (aka it functions like our immune system, but on the smaller bacterial scale, their invaders are viruses or DNA, so the CRISPR system uses the viruses own DNA as a way to attack it back, just as we use antibodies to identify our invaders and attack them back.)
Why Modify B Cells?
That's a fair question to ask. The main goal of modern medicine is to extend the human livespan as long as possible. One way to do that is to give our own bodies the correct tools to target bad material (eg. cancer) for more effective elimination. B cells play an integral role in our adaptive immune system functioning, as they are responsible for both presenting antigens (a marker for a foreign invader) and also producing antibodies, both of which "inform" T Cells who to go after and kill. Research has been done to make modifications to T-cells directly (the most advanced of those therapies is called CAR T and if you are interested in it, you can read a more generalized summary HERE) and these genetic modifications allow them to be more effective fighters against disease.
B cells are also attractive candidates for genetic modification and they have certain advantages over T cells. One advantage is that they are very easy to isolate, and grow very well in a lab setting. So it becomes possible to quickly and easily acquire and grow up very large quantities of cells. Were those cells then modified to express a particular antibody they could become a very effective tool for fighting against cancer.
Some Of The Authors' Work
In this paper the authors were trying to set up conditions necessary to allow for CRISPR manipulation of B Cells, so the work serves as a good example of what researchers need to do early on in the development of a technique such as this. The work isn't the most flashy, but I think this article serves as a great example of how basic science is pushed forward on a topic.
Establishing Growth Conditions For The B Cells
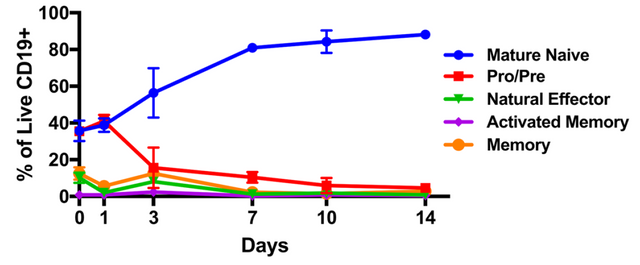
The researchers utilized a protein called CD40L which is a protein responsible for binding to a receptor on the surface of immune cells (both T and B) which results in increased rates of cell growth. Upon treatment of the cells with the CD40L protein, the types of cells in the culture were monitored and the respective proportions reported (as we see in the figure to the left). Here we see that the cells gradually grew to a composition which was about 80% mature native B cells.
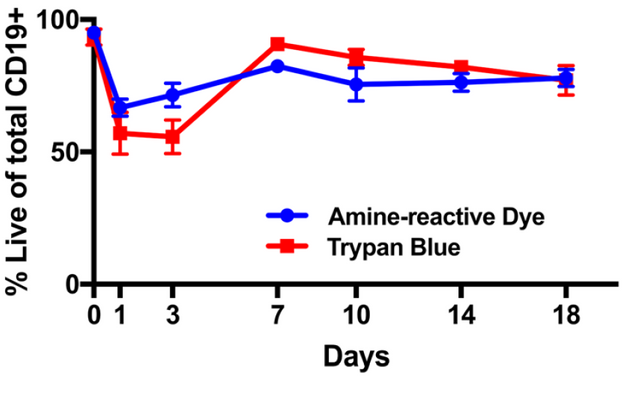
The authors also looked to see what proportion of the cells remained living. To accomplish this they used two dyes, an amine reactive die and trypan blue. Both of these dyes are only able to stain dead cells, living cells do not become colored. Through this they show that over the course of their experiment, their growth conditions result in about 85% living cells (which is good).
Getting DNA/RNA Into The Cells
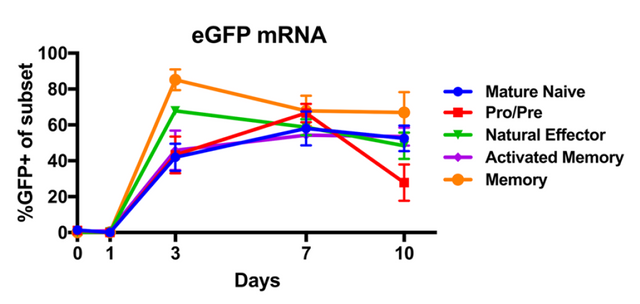
In order to make modifications to the B cells, one has to be able to get nucleic acids (DNA/RNA) into them. We do this through a process called electroporation where cells are zapped with high voltages (here the authors settled on three 10 milisecond pulses of 1400 volts), this weakens the cellular membranes (makes them more porous) and as a result some population of the cells may take up DNA that is in the media around them. Here the authors electroporated with mRNA containing a green fluorescent protein (GFP). They could then monitor the proportion of each variety of B Cell which turned green. We can see that after three days (but not before) in culture, the cells were receptive to the mRNA and it resulted in 40% or greater amounts of cells of each variety expressing the fluorescent protein.
Using CRISPR To Kill A Gene In B Cells
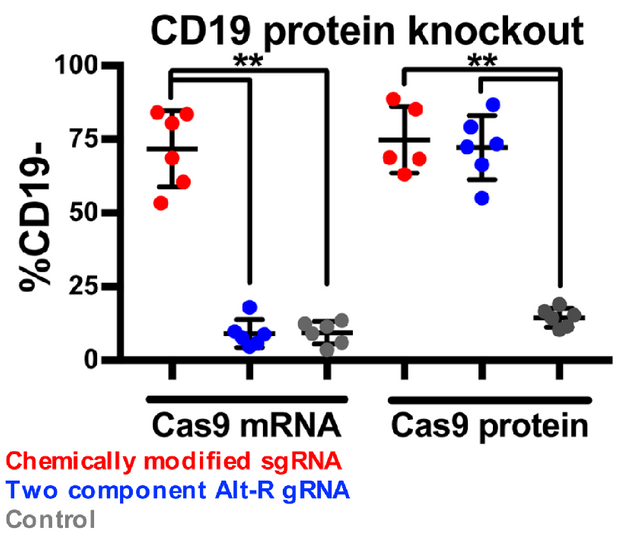
The researchers to this point have established effective culture, and DNA electroporation of the B Cells. Aka they can grow em, and they can get Cas9 and RNA guides in by their electroporation protocol. Now, can they use this to change the expression of a gene? To address this the authors targeted a surface receptor present on the B Cells called CD19.
Here the authors were using fluorescently labeled antibodies to measure the amount of the CD19 cell surface receptor that was expressed after treatment with a few combinations of Cas9 and Guide RNA's. Here they used two different "styles" of guide RNA's and simultaneous or not transfection of Cas9. While I am not going to go into the nitty gritty about these two guide RNA types (chemically modified vs Alt-R). I have included some links to these two methods in case you would like to read more about what they entail.
What we see in the figure to the right, is that successful knockout of the CD19 gene occurs both with simultaneous transfected of the guide RNA and Cas9 nuclease and with electroporation of the guide and direct addition of the Cas9 protein. The authors note that better results were obtained (more cells lived) when using the chemically modified guide RNA and the cas9 protein rather then the cas9 mRNA, so this is the method they will move forward with.
Conclusions
The authors here have established protocols for expression, transfection and use of CRISPR to make edits in human B Cells. Potentially paving the way for an additional avenue of modified immune cell cancer therapies down the line.
- https://www.nature.com/articles/s41598-018-30358-0.pdf
- https://www.cancer.gov/about-cancer/treatment/research/car-t-cells
Thank you, my apologies, I am a dentist, and I am working for the Univerdidad del Zulia, in Venezuela where I gather all the potentials and uses of CRISPR, for that reason I was attracted to its publication. I was a bit severe and rigid and unhappy, with comments hahaha, when I improve my English we will have better communication, thanks again, I leave what should have been my original comment, only to relate what you publish, with what I have been doing research:
CRISPR-CAS9 system will be able to identify the causative genes in many oral pathologies and disorders including early childhood dental caries. Research suggests an association between the distribution of CRISPR sites and the clinical manifestations of caries.
CRISPR will also help in the identification of genes that suppress the tumor-promoting properties of the genes that cause oral cancer. Gene editing technology with its ability to identify, delete or replace genes can improve the prognosis of Squamous cell carcinoma of the oral cavity that arises due to viral and bacterial factors.
Greetings, that this very well, ah, we miss the rest of the alphabet from n to z hahaha
I didn't know this, interesting.
Yep, that is one application certainly. Additionally like what was being discussed here, immuno oncology applications are also on the table, making immune cells attack the oral carcinomas.
I ran out of accounts :D
Hey @aplausos
Here's a tip for your valuable feedback! @Utopian-io loves and incentivises informative comments.
Contributing on Utopian
Learn how to contribute on our website.
Want to chat? Join us on Discord https://discord.gg/h52nFrV.
Vote for Utopian Witness!
One of the major problem faced in subunit vaccines is inability to form a store of long term memory B cells. I was just wondering a few days back that why not just screen for antibodies in-vitro and the CRISPR the B cells derived from pateints with the potent VDJ recombined gene. Then reinject them. Even better if you can differentiate them into memory subtype. This is a very cool paper. Thanks for sharing.
As human beings, we have developed sophisticated immune systems that allow our bodies to fight against viral infections that harm us. Surprisingly, bacteria - despite being unicellular organisms - often possess a similar adaptive immunity, called CRISPR-Cas systems. But these CRISPR-Cas systems of bacteria have a very different function to that of our immune system
One of the least understood aspects of the CRISPR-Cas systems of bacteria is how these microorganisms control their activity, since an excess of this can result in an autoimmune disease, and a shortage of activity could allow the viruses to destroy entire bacterial communities. The team's research shows that by communicating openly with each other, the bacteria reach the right balance between the two results. Greetings, you are very good job in this publication I am the subject
forgive my English written is not very fast and I made a mistake without wanting to transcribe my comment, I think it is not reason to mock, with all respect, is not the spirit of this community, a thousand apologies and that you are very well
Have some fun, live a little :)
Oh no? My sarcasm is what it is.
b
c
d
e
f
g
h
Not sure what you mean here.
The bacteria have a very good ability to not recognize "self" nucleic acids and incorporate them into the CRISPR Array. Though indeed the mechanism is not well understood. In fact one of the least well understood aspects of CRISPR relative to the bacterial immune system itself is the acquisition phase by Cas1 and Cas2.
Nevertheless, the focus of this blog was on the application of CRISPR editing in human B Cells. My initial description of CRISPR was just to get people on the same page, and make a joke about the use of one adaptive immune system to make modifications to another entirely separate adaptive immune system. :)
Great choice of a paper to analyze. It's open access with a clear goal and a focus on relevant modern techniques. However, it is obtusely dense and includes a few extra figures that don't add much to the core story (blame it on being a nature publication). Your summary definitely makes it easier to understand.
Well I was only trying to hit the major points :)
I left out a lot, and certainly some relevant material. I didn't even explain the material in full detail because I don't feel that diving down that far into the weeds is important for a non "expert in the field" audience.
Anyway thanks T.
Greetings! very interesting your post, in fact, recently I was reading some abstract related with tha function of the Natural Killer cells, my best whishes for you,and go ahead with this promising research.
it turns out that antibody cells are very important also for humans, and also need for immunity
I know B cells are implicated in systemic lupus. So far, drug therapies targeting these cells have had limited success. You mention cancer therapies--I wonder, wouldn't this gene modification also potentially be useful in developing drug therapies for lupus?
It could be useful for a variety of issues pertaining to immune functionality yes.
nice right up
Thank you
Nice post I love science thank you for sharing @justtryme90
Posted using Partiko Android
Thanks
By this work, I think CRISPR is such a versatile tool for genetic manipulation for improved immunity.
Thanks for sharing.
@sciencetech
STEM contributor