Could Isomerization of Alkaloids in Common Black Pepper (Piper nigrum) Provide New Leads for Pharmaceuticals?
In one of my past lives I was a medicinal organic chemist using natural products from plants to discover new drugs and formulations. Sadly, the trend in the pharmaceutical industry has been away from unpatentable natural drugs to more profitable synthetic compounds.
My philosophy today is the antithesis of that particular past life: As a senior citizen I am convinced that exercise, nutrition and minimal medications, with selective natural remedies where appropriate, are the best course for one's healthcare.
Still, I Can't Help but Think About Molecules
Black Pepper, a.k.a. "The King of Spices" has been prized for centuries (Wikipedia) as a condiment and medicine. I eat larger amounts of ground pepper and raw peppercorns than the average American, I'm sure.
Recently I was reading a 2016 paper by Gorgani et. al. discussing a review of the chemical properties and medicinal uses of piperine, the active principal in black pepper responsible for its pungency.
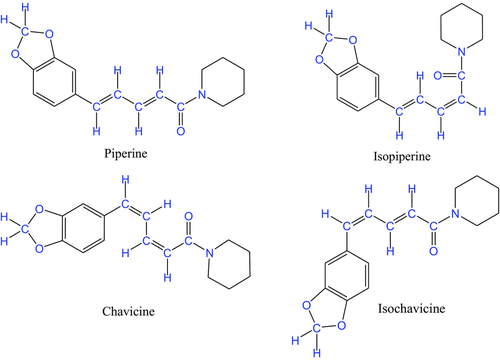
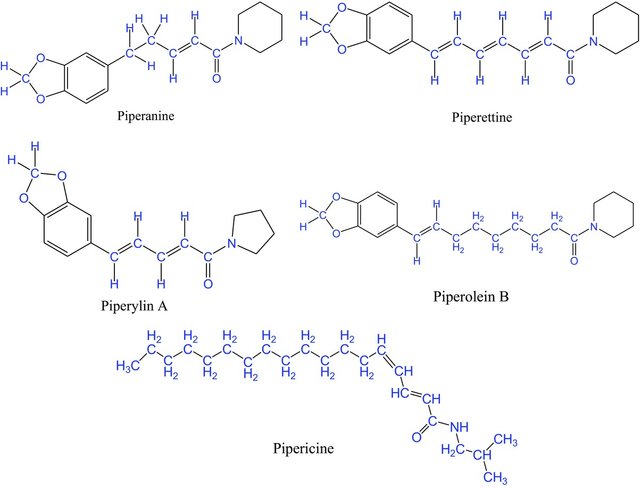
Piperine, isopiperine, chavicine, isochavicine, and other similar compounds are known to occur in black pepper, and their medicinal and culinary values are ancient knowledge.
I remember isolating piperine from pepper by extraction with chloroform as one of my class experiments learning to isolate natural products.
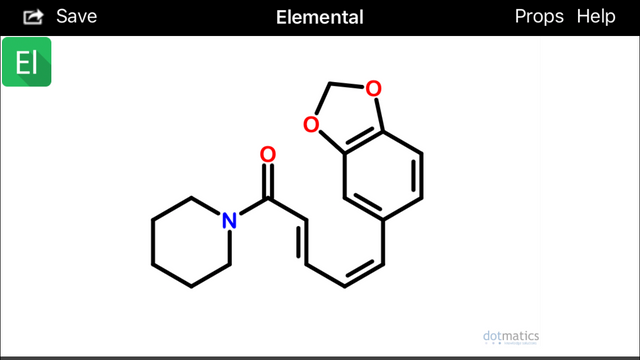
Isochavicine May be a Lead
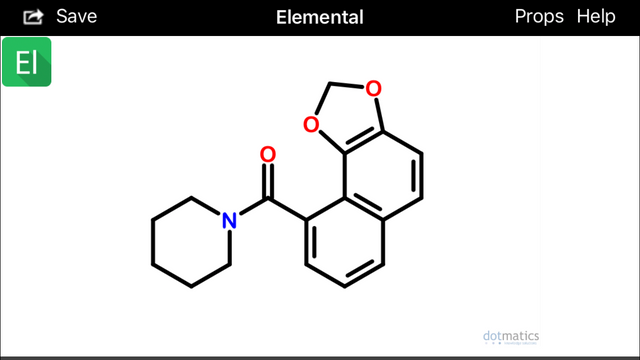
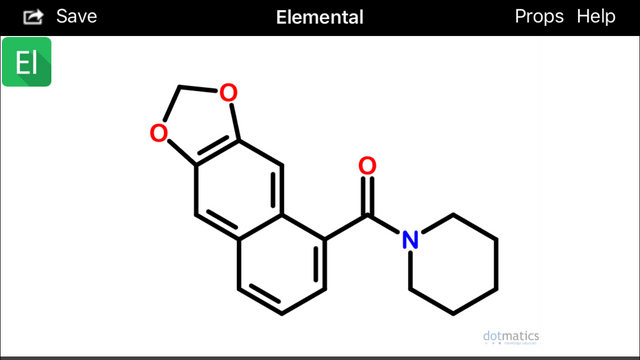
If you look at isochavicine with rotated 3,4 (beta, gamma) bonds, at least two interesting conformations become apparent:
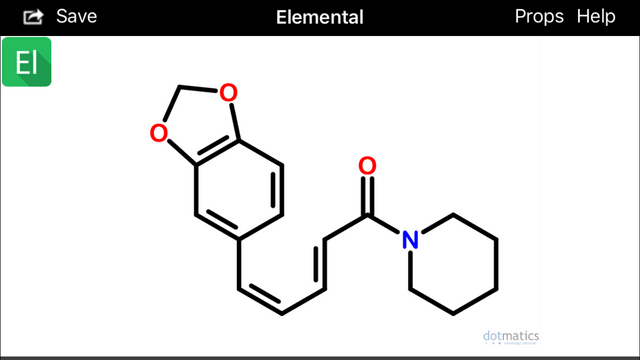
These configurations (above) would hypothetically be extremely prone to spontaneously undergo sigmatropic Diels Alder cyclization and aromatization (source) to form two possible alpha(1)-napthoyl-piperidine amide derivatives (below):
Diels-Alder cyclization or other photocatalytic conversions could occur in vivo after absorption and within skin cells exposed to sunlight (Wikipedia).
Biological Activity?
It is likely some type of biological activity of medicinal advantage could be derived from these compounds. The conjugated double bonds between a carbonyl and an aromatic group could undergo a wide range of reactions to create derivatives.
The cis, cis isomer, chavicine, is sterically hindered from adopting these conformations due to the planar arrangement of conjugated double bonds, but synthetic analogs might be possible.
Such derivatives are not known to occur in nature, so they could be patented. Furthermore they are conformational isomers of naturally-occurring biologically active compounds with potential for further investigation.
Congratulations @qiyi! You received a personal award!
Click here to view your Board
Congratulations @qiyi! You have completed the following achievement on the Steem blockchain and have been rewarded with new badge(s) :
Click here to view your Board
If you no longer want to receive notifications, reply to this comment with the word
STOPWoo! Love your post - we've given you an upvote with a chance to be featured in the weekly curation. We’ve also resteemed it. We encourage you to use the #naturalmedicine tag so we can more easily find you.
If you're a supporter of all things natural healing, and haven't already got on board our collective, you might like to read our introductory post here. We'd also love to welcome you on Discord here
Great article! I was just in Costa Rica where I visited a spice plantation growing black pepper, vanilla, cacao and other spices organically.
I'll put that on my bucket list! I remember Baltimore Harbor where the McCormick Spice Co. had it's port, you could smell the aromas.
Short but deep article. I also worked in "my past life" with various natural products. It's a strange passion.
I don't want cars, boats, or golf equipment...I need a lab (or computer and platform) to use my skills.
Me too. Lab all the way and I am building one. I will share my notes sir!
I've designed and started several labs and programs. I look forward to your posts.
Love the post, I consume a hell of a lot of black pepper freshly ground as well but never raw, I would be interested in trying it! I use it in combination with turmeric since the black pepper compounds increase the potency of the turmeric. You’re not the only one who uses it so much around here! ;)
Posted using Partiko iOS
This post has been voted on by the SteemSTEM curation team and voting trail in collaboration with @utopian-io and @curie.
If you appreciate the work we are doing then consider voting all three projects for witness by selecting stem.witness, utopian-io and curie!
For additional information please join us on the SteemSTEM discord and to get to know the rest of the community!
Wow, thanks!
Hi @qiyi!
Your post was upvoted by Utopian.io in cooperation with @steemstem - supporting knowledge, innovation and technological advancement on the Steem Blockchain.
Contribute to Open Source with utopian.io
Learn how to contribute on our website and join the new open source economy.
Want to chat? Join the Utopian Community on Discord https://discord.gg/h52nFrV
Thank you!
Great read! Love the high-quality science-based natural product content. Pepper is definitely a spice that we could not live without. We just gave you a small upvote together with our many followers of the @nunesso blog. Have you heard of @nunesso? If you write more content like this be sure to use the #nunesso or #naturalproducts tag. We are looking to reward authors and content creators who produce exceptional natural product-related articles to give upvotes, resteems and be featured in our curation posts!
Thanks for making me aware of your tag and blog. I find interesting info and will follow for more.