An ABC (or rather an alpha-beta-gamma in Greek) of the origins of the chemical elements
Standard cosmology is today well motivated by all observations. It includes in particular a mechanism named big bang nucleosynthesis that describes how the lighter chemical elements (hydrogen, helium, lithium and beryllium) have been produced.
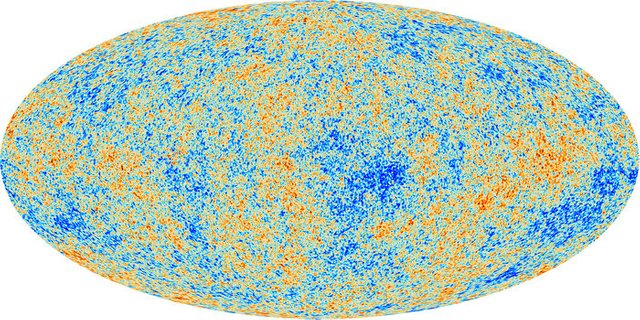
[image credits: ESA]
Big bang nucleosynthesis, that moreover correctly explains the relative proportion of the elements in the universe, was initially worked 70 years go out by Alpher (a PhD student at that time) and his advisor Gamow.
There are of course differences between today’s standards and the initially-proposed work, but the bulk of it were there.
The physicist Bethe also signed the corresponding publication, so that its author list is Alpher-Bethe-Gamow. This almost matches the first three letters of the Greek alphabet, alpha-beta-gamma. Consequently, this article is known today as the alphabetical article.
THE EARLY UNIVERSE HISTORY - INFLATION
The inflation epoch matches the very first moments of the life of our universe. By very first moments, I actually refer to a tiny amount of time equal to roughly 0.000000000000000000000000000000001 second.
At that moment, the universe exponentially expanded before continuing at a more relaxed pace. Even if the expansion of the universe is accelerating today, this is in no way as brutal as what happened during the inflation epoch.
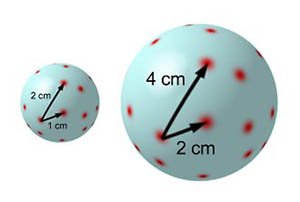
[image credits: hk-phy and everywhere on the web]
A funny way to visualize the expansion of the universe is to take a balloon and draw red dots on it to represent the stars and the galaxies.
When inflating the balloon, the dots are getting more and more separated from each other (see on the right).
But let us go back to the very beginning of inflation.
The universe is usually referred to as a cosmic soup. It is very hot, so that particle-antiparticle pairs can be created from the vacuum. The inverse reaction also occurs, at an equal rate: particles and antiparticles annihilate.
The universe is thus in thermal equilibrium: the net product of both reactions is zero.
THE EARLY UNIVERSE HISTORY - BARYOGENESIS
After inflation, the universe goes on with its expansion, and its temperature decreases. Certain processes leading to the creation of a particle-antiparticle pair consequently become forbidden as the energy is too diluted. For these particle species, only the annihilation process remains.

[image credits: NASA]
These particles have therefore almost all disappeared today, and one needs powerful accelerators to recreate them.
There is nevertheless a catch. Although an equal amount of particles and antiparticles are destroyed, a tiny matter-antimatter asymmetry appeared.
The asymmetry must be generated by some mechanism called baryogenesis (more details one day in another post).
Nevertheless, the universe ends up to be matter dominated and there is almost no trace of antimatter anymore.
BIG BANG NUCLEOSYNTHESIS
In the previous paragraph, we described the first 10 seconds of the universe life. The universe contains bunches of protons and neutrons, but no atomic nuclei yet. Electrons are also present, and photons (or radiation) dominate the energy budget of the universe.
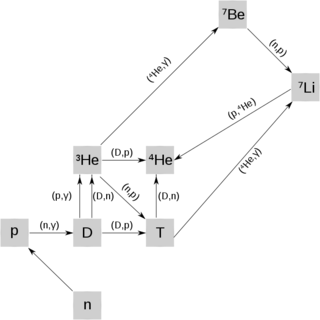
[image credits: Wikipedia ]
The mechanism behind the formation of the lighter nuclei is known as Big Bang nucleosynthesis. This says that all atomic nuclei are produced by successive neutron captures.
After a few minutes, the universe is sufficiently cool for deuterium nuclei (made of one neutron and one proton) to be formed (and not destroyed by the surrounding photons).
Starting from there, the surrounding neutrons allow for the formation of tritium (1 proton and 2 neutrons), helium-3 and helium-4 (2 protons and either 1 neutron or 2 neutrons), beryllium-7 (4 protons and 3 neutrons) and finally lithium-7 (3 protons and 4 neutrons).
THE ALPHABETICAL ARTICLE
The first proposal explaining the origins and the abundance of the lightest elements were done by a PhD student named Alpher in 1948, together with his advisor Gamow.
They suggested that all atomic nuclei are produced by successive captures of a single neutron, as already said above. This however fails for any heavy nucleus with more than 7 nucleons (a nucleon is a generic wording for protons and neutrons).
No heavier element can indeed be formed because there does not exist any stable nucleus with 5 and 8 nucleons. Intermediate steps are thus missing. I indeed recall that in the proposal of Alpher and Gamow, one can only capture one single neutron at a time.

[image credits: pixabay]
For the heavier guys, stars are thus necessary. These are denser objects in which triple helium collisions can occur and produce carbon. This however happened much much later.
Alpher and Gamow have names mimicking very closely two Greek letters: alpha (Alpher) and gamma (Gamow). Gamow consequently asked his friend Bethe (beta) to co-sign the paper.
The reason is trivial: physicists are funny people who like jokes.
Alpha-beta-gamma is indeed better than alpha-gamma. After joining the collaboration, Bethe helped in preparing the manuscript and started to work on big bang nucleosynthesis.
SUMMARY AND REFERENCES
In this post, I briefly described the life of the early universe. A few seconds after the big bang, chemical elements could start being produced from the protons and neutrons present in the universe at that time.
The first scientific article detailing such a production mechanism is now as the alpha-beta-gamma or the alphabetical article.
It was written by Alpher, Bethe and Gamow and describes big bang nucleosynthesis. Their proposal were found to describe in success the relative abundance of 99% of the visible mass of universe (the few lightest elements), but failed for the heavier element that are connected to stars. It is a great success, isn't it?
More information can be found in the alpha-beta-gamma article, available for free, as well as in these lecture notes on baryogenesis and these lecture notes on inflation.
thanks for sharing
interesting publication friend, it is always good to know a little history and more if it is the chemical elements
^^
hello friend, I hope you are well, greetings
Hello Friend how are you? I have a question for you? this morning I have just reviewed the publications that I have made since last week and I realized that most of the votes are in 0% I really do not understand what is due, because my publications are real stories made by my people and the images and theory of the content of the disease are all with their respective references, I hope you can clarify this because I do not know what happens @lemouth
You have been found copy pasting from Spanish sources. For this reason, some votes may have disappeared. That's it :)
thank you very much for clarifying my doubt, I already learned of my error this time I will publish in a more original way I promise
i'm interested in the ideas of exogenesis/panspermia, for me it seems like the obvious theory at this point. but the mystery is where did it all come from/be created to get to earth in the first place. therefore we are all really aliens... but really not when you view the whole universe as our home.
What always plays a role here in this stuff is the faith / religion aspect also.
It will always ruffle feathers when dealing with these types of science issues.
Panspermia is an interesting idea too. It is however not really my field. I really discuss here the origins of the elements as elements. Purely chemicals :)
Exactly! :)
What are the odds for the Alpher Bethe Gamow to have such publications haha...
This depends how you quantify Gamow's humour? :)
I never knew how these chemical elements originated. We were only shown their positions in the periodic table, and their physical and chemical properties. Now my knowledge has been broadened. Thanks for sharing
They must come from somewhere, don't they? :)
Thanks for your comment ^^
Nice post @lemouth. Thanks for sharing.
My pleasure :)
Congratulations @lemouth! You have completed some achievement on Steemit and have been rewarded with new badge(s) :
Click on any badge to view your own Board of Honor on SteemitBoard.
For more information about SteemitBoard, click here
If you no longer want to receive notifications, reply to this comment with the word
STOP