#cold-drinks-Deep analysis report,A threat to you!
hello steemians ! this is the report that is used to prepare for my college project so i just post it also for you!

Introduction:-
The era of cold drinks began in 1952 but the indianization of industry marked its beginning with launching of limca And goldspot by parley group of companies. Since, the beginning of cold drinks was highly profitable and luring, many multinational companies launched their brands in India like pepsi and coke. Now days, it is observed in general that majority of people viewed Sprite, Miranda, and Limca to give feeling of lightness, while Pepsi and Thumps Up to activate pulse and brain.
Theory:-
Cold drinks of different brands are composed of alcohol, carbohydrates, carbon dioxide, phosphate ions etc. These soft drinks give feeling of Warmth, lightness and have a tangy taste Which is liked by everyone.
Carbon dioxide is responsible for the formation of froth on shaking the bottle. The carbon dioxide gas is dissolved in Water to form carbonic acid which is also responsible for the tangy taste. Carbohydrates are the naturally occurring organic compounds and are major source of energy to our body. General formula of carbohydrates is CX (H20)Y.
On the basis of their molecule size carbohydrates are classified as:-
Monosaccharide, Disaccharides and Polysaccharides. Glucose is a monosaccharide with formula C6H1206 . It occurs in Free State in the ripen grapes in bones and also in many sweet fruits. It is also present in human blood to the extent of about 0.1%. Sucrose is one of the most useful disaccharides in our daily life. It is widely distributed in nature in juices, seeds and also in flowers of many plants. The main source of sucrose is sugar cane juice Which contain 15-20 % sucrose and sugar beet which has about 10-17 % sucrose. The molecular formula of sucrose is C12H2201l. It is produced by a mixture of glucose and free dose. It is non-reducing in nature Whereas glucose is reducing. Cold drinks are a bit acidic in nature and their acidity can be measured by finding their pH value. The pH values also depend upon the acidic contents such as citric acid and phosphoric acid.
Types of Acid:-

Most soft drinks contain one or two common food acidulants — phosphoric acid and citric acid. Occasionally, other acidulants such as malic acid or tartaric acid are also used. Animal studies have shown that phosphoric acid is very erosive at pH 2.5 but much less so at pH 3.3. Citric, malic and tartaric acids are considered to be especially erosive because of their acidic nature and the ability to chelate calcium at higher Ph (48).
Citric acid was more erosive than malic acid when formulated to experimental drinks at high pH (23, 52). In an in vitro experiment W/est etal. (53) concluded that citric acid caused far more erosion over the pH range employed than phosphoric acid for enamel and dentine. Citric acid was found to compare with hydrochloric acid highlighted dissolution and chelation effects. Phosphoric acid caused minimal erosion over pH 3 for enamel and pH 4 for dentine.
For Determination of content of cold drinks
APPARATUS
Test tube holder
Test tube stand
Stop Watch
Beaker
Burner
PH paper
Tripod stand
China dish
Wire gauge
Water bath
CHEMICALS REQUIRED
Iodine solution
Potassium iodine
Sodium hydroxide
Fehling’s A & B solution
Linle Water
Concentrated HNO3
Benedict solution
Ammonium molybdate
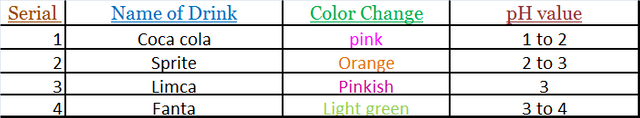
Detection of PH:-
• 1-2 drops of the sample of cold drink of each brand Was taken and put on the pH paper.
• The change in the color of pH paper was noticed and was compared with the standard pH scale.
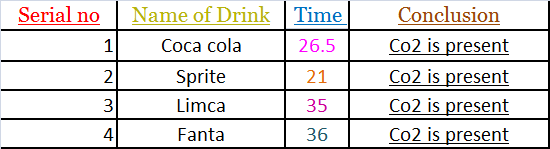
Test for Carbon Dioxide:-
EXPERIMENT
As soon as the bottles were opened, one by one the sample was passed
through lime water.
The lime Water turned milky.
INFERENCE
All the soft drinks contain dissolved carbon dioxide in water. The carbon
dioxide (CO2) dissolves in Water to form carbonic acid, which is responsible
for its tangy taste.
CHEMICAL REACTION INVOLVED
Ca(OH)2 (S) + CO2(g) --------- --9CaCOs (S) + H2O(s)
Test for glucose:-
Glucose is a reducing sugar acid. Its presence is detected by the following
test:-
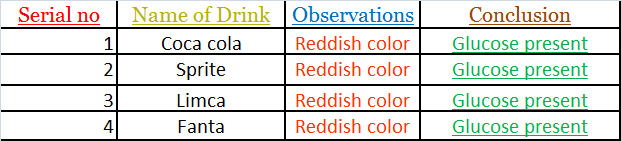
- BENIDICTS’S SOLUTION TEST:-
• A small sample of cold drink of different brands Was taken in a test tube and a few drops of Benedict’s reagent were added.
• The test tube was heated for few seconds.
• Fonnation of reddish color confirms the presence of glucose in cold drinks.
Observations:-
INFERENCE
All the samples gave positive test for glucose With Benedict’s reagent.
Hence all the drinks contain glucose.
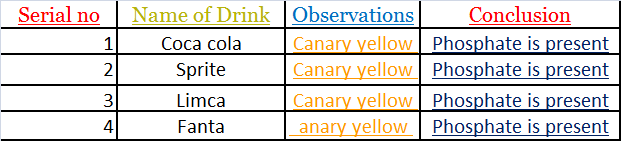
Test for Phosphates:-
Sample of each brand of cold drink was taken in a separate test tube and ammonium molybdate followed by concentrated nitric acid (HNO3) was added to it.
The solution was taken heated and the color of the precipitate confirms the presence of phosphate ions.
CHEMICAL REACTION INVOLVED
NaHPO4 + 12 (NH4)2MoO4 + ZIHNO3 +3H+ --------- ---) (NH4)3PO4.l2MoO3
+2lHN4NO3 +12H2O
INFERENCE
All the soft drinks contain phosphate ions which are detected by the
presence of phosphate when canary yellow obtained.
Observations:-
Test for Alcohol:-
Samples of each brand of cold drinks are taken in sample test tube and iodine followed by potassium iodide and sodium hydroxide(NaOH) solution is added to each test tube.
Then the test tube are heated in hot water bath for 30 minutes yellow colored precipitate confirmed the presence of alcohol in cold drinks
Observation:-
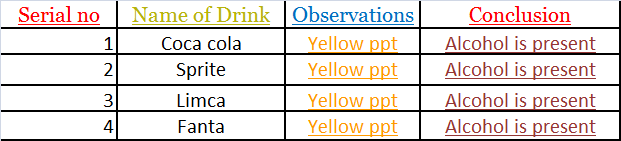
INFERENCE
All the Brands of Cold Drinks Contain Alcohol.
CHEMICAL REACTION INVOLVED
CH3CH2OH +4I2+ 6NaOH ---- --9CHI3 + HCOONa +5NaI +5H2O
Test for Sucrose:-
• 5 ml samples of each brand of cold drinks was taken in a china dish and heated very strongly until changes occur.
• Black colored residue left confirms the presence of sucrose in cold drinks.
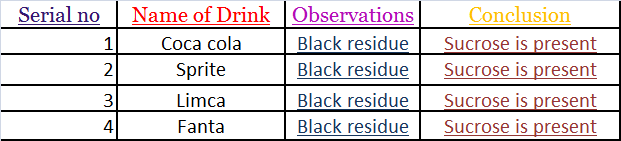
INFERENCE
All the brands of cold drinks contain sucrose‘ But amount of sucrose varies
in each brand of drink. Fanta contained highest amount of sucrose.
Result:-
After conducting several tests, it was concluded that the different brands of
cold drinks namely
• Coca cola
• Sprite
• Limca
• Fanta
All contains glucose, alcohol sucrose, phosphate, ions and carbon dioxide.
All are acidic in nature.
On comparing the pH value of different brands coca cola is most acidic and limca is least acidic of all the four brands taken.
pH value of coca cola is nearly equal to disinfectant which is harmful for body.
CARBON DIOXIDE
AMONG THE FOUR SAMPLES OF COLD DRINKS TAKEN —SPRITE HAS MAXIMUM AMOUNT OF DISSOLVED CARBON DIOXIDE AND FANTA HAS MINIMUM AMOUNT OF DISSOLVED CARBON DIOXIDE.
DISADVANTAGES OF COLD DRINKS

soft drinks are little more harmful than sugar solution. As they contain sugar in large amount which cause “diabetes”.
Soft drinks can cause weight gain as they interfere with the body’s natural ability to suppress hunger feeling.
Soft drinks have ability to dissolve the calcium so they are also harmful for our bones.
Soft drinks contain “phosphoric acid” which has a pH of 2.8. So they can dissolve a nail in about 4 days.
For transportation of soft drinks syrup the commercial truck must use the hazardous matter place cards reserved for highly consive material.
Soft drinks have also ability to remove blood so they are very harmful to our body.
USES OF COLD DRINKS
• Cold drinks can be used as toilet cleaners.
• They can remove rust spots from chrome car humpers.
• They clean corrosion from car battery terminals.
• Soft drinks are used as an excellent ‘detergent’ to remove grease from clothes.
• They can loose a rusted bolt.
Carbonation of beverages
In one study that compared acid clearance of noncarbonated and carbonated soft drinks in the mouth, there was no statistical difference in the clearance between the drinks. It was concluded that carbonation may not directly responsible for the erosive potential of different beverages (73).
Cola and non-cola beverages Some in vitro studies have shown that 50/a drinks, despite having the lowest pH on opening, were easier to neutralize than the fruit juices and non-cola drinks (34, 74, 75), while Jain et al. (76) concluded from their study that non—cola drinks were more erosive than cold drinks. The type of acid has been used to explain the ability of the non-cola drinks and fruit juices to resist pH change; phosphoric acid is normally used in the cola drinks while citric acid predominated in the fruit juices and the non—cola drinks (7 Surprisingly, an in vitro study has shown that citric acid caused far more erosion than phosphoric acid (53).
Please upvote or comment
Congratulations @thaps2001! You have completed the following achievement on Steemit and have been rewarded with new badge(s) :
Click on the badge to view your Board of Honor.
If you no longer want to receive notifications, reply to this comment with the word
STOPDo not miss the last post from @steemitboard:
SteemitBoard and the Veterans on Steemit - The First Community Badge.