Metrology: The Science of Measurement and how it has shaped the course of History #2.
Hello, great people. I'm back here once again to write a sequel to my last post on metrology. you can quickly have a glance here. Today, just like I promised, I'll be discussing on the conversion method of changing units of measurement from one physical quantity to another and also be going back in time into history to explain the three fundamental quantities in physics i.e mass, length and time.

Source: CGKilogram.jpg, Author: User:Greg, CCBY-SA 3.0
Conversion method of changing units
In a measurement, we frequently need to change the units in which our quantities are expressed and to do this, we may need to employ a method known as the "chain-link conversion". This method is applied by multiplying the original measurement we want to convert by a conversion factor which is a ratio of units that is equal to 1. Let's look at an example to understand what I'm saying in essence. Since we know that 1 meter and 100cm are same length intervals, they can be written as:
1 m/100 cm = 1 and 100 cm/1 m = 1
Then, the ratio (1 m)/(100 cm) and (100 cm)/(1 m) can be used as a conversion factor, which is very much different to writing just 1/100 = 1 or 100 = 1 without including the units. Meaning that the numbers, as well as the units, are very crucial and each one of them must be considered or regarded together. When we multiply any quantity by 1 ( i.e unity), the answer gives back the quantity without any change. The conversion factors are very useful to eliminate unwanted units in a chain-link conversion method. Let's take, for instance, we are to convert 7 meters to centimeters, and since we already know that in every 1m, there's always a 100cm; then it can be solved as:
7 m = (7 m)(1) = (7 m)(100 cm)/(1 m),
7 × 100 = 700 cm
Let's take two more examples before we move ahead. Assume we want to convert 95 Å (pronounced as Angstrom) to centimeter. Here are the steps; we must first know the equivalent of 1 Å to meters which = 10-10 m, meaning that 95 Å is equal to 95 × 10-10 m, but we know that 1 m is equivalent to 100 cm. Then we can apply the conversion factors of 1 m/100 cm = 1 and 100 cm/1 m = 1 and can thus convert 95 Å to cm as thus:
95 Å = (95 × 10-10 m)(1) = (95 × 10-10 m)(100 cm)/(1 m),
95 × 10-10 × 100 cm = 0.00000095 cm = 9.5 × 10-7 cm.
On the last example, what if we are working with pressure and need to convert from one pressure scale to another. Let's take, for instance, we want to convert 2100 millimeter-mercury (mmHg) to Atmosphere (atm). Normally, 760 mmHg = 1 atm and which can be written as a conversion factor as 1 atm/760 mmHg = 1 and 760 mmHg/1 atm = 1. Then, we can go ahead and convert 2100mmHg by:
7 m = (2100 mmHg)(1) = (2100 mmHg)(1 atm)/(760 mmHg),
(2100/760) atm = 2.76 atm
In a situation whereby the conversion factor you used don't cancel out the undesirable units, then reverse the factor and attempt it again.
Since the understanding of units changing from one physical quantity to another has been clearly understood, then let's delve into the history of the three most common and popular fundamental physical quantities. Mind you, we have up to seven fundamental quantities which are the length, mass, time, temperature, current, amount of substance, and luminous intensity. Of which I'll be doing most of the exposition on the first three aforementioned physical quantities.
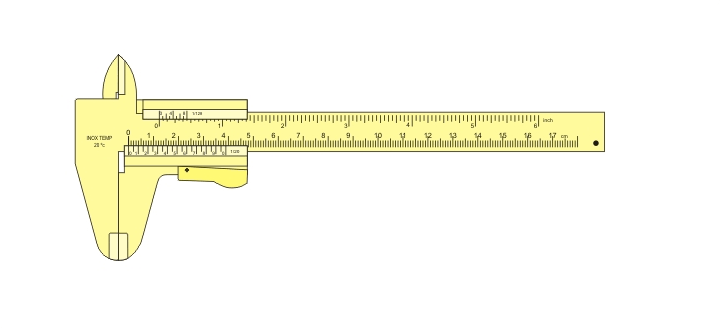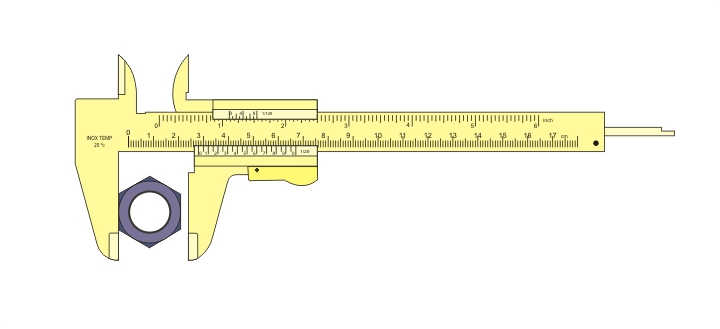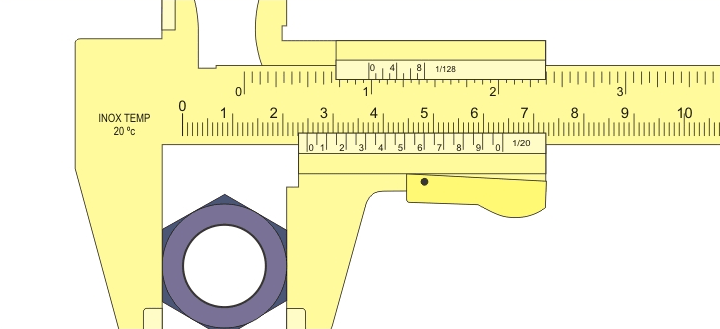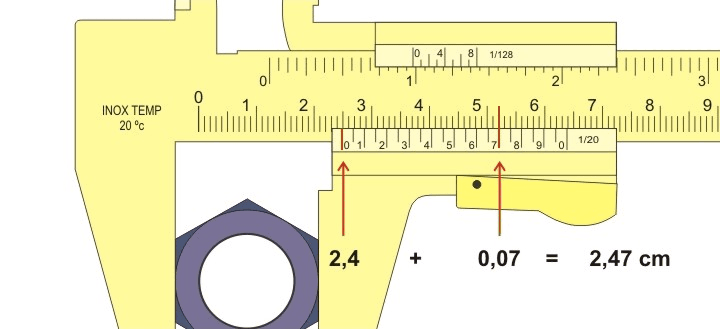
Author: Joaquim Alves Gaspar, CC BY-SA 2.5
Length
In the year 1792, the word "meter" was born. It was the result of the new weights and measures system that was newly adopted back then in France. The meter was defined to be an equivalent to 1/10,000,000th (one ten-millionth) of the distance it takes to go to the north pole from the equator. Some years later, another definition for meter arose as the distance between two lines that are drawn very close to the end of a platinum-iridium bar which was then taken as a standard meter bar. As time goes by, people started demanding for a more accurate and precise measuring standard. This need eventually resulted in the discovery of a new standard for the meter in 1960.

Author: NIST website, Public Domain
This new standard was founded on the wavelength of light. It was concluded that one meter is equal to 1,650,763.73 wavelengths given off by a Krypton atom contained in a glass receptacle. And it was taken that it can be practicalized as such anywhere on the surface of the earth. Some 20 years afterward, people needed a more accurate and precise standard for measuring length. And that great urge resulted in the meter being redefined as the distance traveled by light in a known time interval. It was then decided at The 17th Conférence Générale des Poids et Mesures (also translated as The General Conference on Weights and Measures), that the meter is the extent of a path traveled by light in a vacuum at a time gap of 1/299,792,458 of a second source
This time gap was chosen as a result of the high precision in the speed of light to be 299,792,458 (2.99 × 108) m/s. And because of that great accuracy in the speed of light, it was well accepted to use as a standard to re-establish the meter.
Time
Time is very important in a man's life. A man will always want to know the time of the day so as to plan his schedules. And if that's the case, then any time standard should be able to answer the question of "what is?" and "when?" an occurence happened.
Any occurence or happenings that continue to reoccur can be considered as a time standard. An example is in the rotation of the earth which takes place every 24 hours a day. Through this behavior of the earth, a length of the day can be known and the concept itself has been in use in that form for centuries back.
This diagram below shows an original example of a watch based on the rotation of the earth. Though the watch has a limitation which is in the way it's been calibrated. If you look very closely, it can be observed that the the hour dial was structured in such a way as to provide a 10-hour day while their is a small dial that's keeping a normal 12-hour time.
Author: De facto, CC BY-SA 4.0
The idea wasn't that correct because the watch was calibrated in line with the rotation of the earth but the calibration can't be used with the precision and accuracy demanded by the modern science and engineering technology. So, to achieve the aim of a better time standard, an atomic clock was developed.
There is an atomic clock at the National Institute of Standards and Technology (NIST) in Colorado, which is now the standard for Coordinated Universal Time (UTC) in the United Statessource. The time signals can be gotten through short wave radio stations. The other relevant information about the time signals can also be gotten at the United States Naval Observatory at their website source. So, before you can set a clock and get an accurate time at your position, you must first put into consideration the time it will take for the time signal to get to you.
Author: NIST, public domain
Also, in the year 1967 at the 13th General Conference on Weights and Measures, the atomic clock (which was based on caesium) was accepted as the standard clock for measuring time and the second was defined as the time it will take 9,192,631,770 oscillations of the light produced by a caesium atom.source One of the most interesting facts about atomic clocks is that they are very reliable and constant to an extent that two caesium clocks can run up to 6,000 years before their readings could vary for more than a second. Even such accuracy can't be seen in the modern clocks that are used nowadays. Finally, let's look into the world of mass......
Mass
The Standard Kilogram
The platinum–iridium cylinder was internationally agreed upon to be the SI standard of mass (i.e equivalent to 1 kilogram). The original sample of the standard is kept at the International Bureau of Weights and Measures (BIWM) very close to Paris. Many exact copies of it were made and sent across the world to be used as a measure in determining the unknown masses of other bodies. An accurate copy of the platinum-iridium cylinder can also be found in the vault of the National Institue of Standards and Technology (NIST), United States.
Author: NIST, public domain
The Second Standard of mass
It was found that it is more accurate to weigh up the masses of atoms with one another than to compare them with the standard kilogram. So, because of that reason, a second standard for mass was internationally agreed upon and that is the carbon-12 atom. The carbon-12 atom is having an equivalent mass of 12 atomic mass units (a.m.u.), where;
1 a.m.u = 1.660 538 86 × 10-27 kg
From this, scientists can now decide the masses of other atoms with respect to the mass of carbon-12.
Conclusion
Measurements and units are very essential in the life of human without which life would have been unbearable to live. How can we do without weighing the mass of objects in our environment? How could we have built so many wonderful things of life without measurements and dimensions; the great pyramids in Egypt, Eiffel tower, airplanes, trains and electrical appliances wouldn't have come to being with measurements. Finally, if not for science of measurements, it would have been difficult to calculate the time difference between different zones.


.jpg)



Congratulations! This post has been upvoted from the communal account, @minnowsupport, by emperorhassy from the Minnow Support Project. It's a witness project run by aggroed, ausbitbank, teamsteem, someguy123, neoxian, followbtcnews, and netuoso. The goal is to help Steemit grow by supporting Minnows. Please find us at the Peace, Abundance, and Liberty Network (PALnet) Discord Channel. It's a completely public and open space to all members of the Steemit community who voluntarily choose to be there.
If you would like to delegate to the Minnow Support Project you can do so by clicking on the following links: 50SP, 100SP, 250SP, 500SP, 1000SP, 5000SP.
Be sure to leave at least 50SP undelegated on your account.
This post has been voted on by the steemstem curation team and voting trail.
There is more to SteemSTEM than just writing posts, check here for some more tips on being a community member. You can also join our discord here to get to know the rest of the community!
Hi @emperorhassy!
Your post was upvoted by utopian.io in cooperation with steemstem - supporting knowledge, innovation and technological advancement on the Steem Blockchain.
Contribute to Open Source with utopian.io
Learn how to contribute on our website and join the new open source economy.
Want to chat? Join the Utopian Community on Discord https://discord.gg/h52nFrV
Great job done on the chain conversion
NmNicely laid out example for even a primary school to understand
Hi, @lawkay
Thanks for coming by and also for the nice comment.
Resteemed your article. This article was resteemed because you are part of the New Steemians project. You can learn more about it here: https://steemit.com/introduceyourself/@gaman/new-steemians-project-launch