Diamonds in the sky - Part 2: Clathrate Stars
In the very far future, our sun will probably end up has a massive spherical diamond the size of a planet. This is what we discussed in the first article. In this paper, I go further, and blend this idea with some of my PhD work which dealt with a special type of crystalline structure: Clathrates.

After a short summary of part 1 in order to place the reader in context, I discuss about clathrate crystalline structures. Yes I know, it is a solid state chemistry topic, not an astrophysics one. Please bear with me: blending subjects which appear at first sight quite distant from one another allows the blooming of stunning ideas.
And that is what I do in this article: blend the idea of elder white dwarf having their crust progressively crystallize as they cool down with what happens when high pressure and high temperature is applied to column 4 elements of the periodic table like carbon, silicon or germanium. The crystallization process leads to a diamond structure, but what if there are impurities present?
This is again another feature that makes Nature amazing...

1/ White Dwarf stars (Summary of Diamonds in the sky - Part 1)
In the first article, we learned that a star fuses hydrogen into helium and uses the energy of this reaction to counter the crushing effect of gravity. Once the hydrogen consumed, and if the star is more massive than half the mass of our sun, helium can in turn fusion into carbon, and even a give out a little oxygen in the process.
The temperature of the core is so high that the outer layers of the star are pushed away providing a beautiful site which is called a planetary nebular. In the center is present a very dense and hot core, a white dwarf.

Once all fusion reactions stop, because of lack of a sufficient concentration of remaining helium reactant, the white dwarf remains stable thanks to a quantum phenomenon called electron degeneracy pressure. At that point, the core is still very hot and composed of a plasma of carbon nuclei with traces of helium and maybe oxygen, all immersed in a sea of degenerate electrons.
Progressively, with time, the white dwarf cools down, starting from the surface. When the surface reaches temperatures lower than 4000K, crystallization of its crust is expected to occur, a little like when liquid water transforms to crystalline ice when cooled progressively down to 0 degrees Celsius. A white dwarf with a surface temperature at 3900K was found recently suggesting that this process may already be happening even if our universe is still relatively young [source]. 
I ended the previous paper on the hypothesis that the conditions of crystallization of the carbon plasma is such that the crust of a cool white dwarf star must be composed mainly of carbon with a diamond crystalline structure. With time, the star cools further and the crystallization process dives deeper into the core: the whole star ends up as a huge spherical diamond floating in the sky. Our sun, in a far future, could well end up as a diamond the size of a planet!
Now, I am going to explore this idea further, and propose a bold hypothesis based on what I have been working on during my PhD: the outer crust of these stars could exhibit instead a carbon clathrate structure.

2/ What is a clathrate structure ?
Under high pressure, carbon crystallizes with an sp3 configuration. That means that the carbon atoms link to each other by forming a tetrahedron.
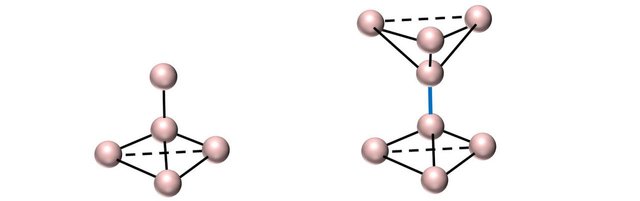
In a sp3 crystal, two tetrahedra connect via a common bond (in blue) which acts as a rotation axis: The tetrahedra can orient themselves relatively to each other by rotation.
In nature, two configurations occur that we can observe by looking along the rotation axis. Either the atoms at the front ‘hide’ the atoms of the back (eclipsed configuration), either the atoms of the back have rotated by 60 degrees, and are now visible when looking along rotation the axis (offset configuration).
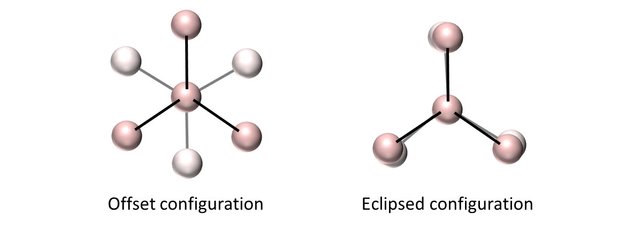
The relative orientations of the tetrahedra will define the overall crystalline structure.
If the configuration is offset, the tetrahedra create a zigzagging network of carbon atoms that fill up all space optimally, thus resulting in a diamond structure.

If the configuration is eclipsed, the crystal formed results in big empty cages that fill up all space and where other atoms can be intercalated. These cages are Euler polyedra, that means that the sum of the number of vertices (N) and the number of sides (S) will be equal to the number of edge (E) plus 2:
N + S = E + 2.
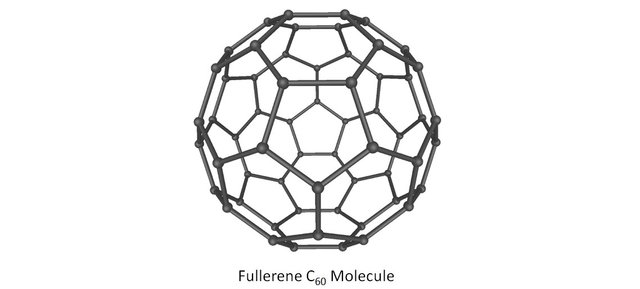
As an example, the Buckminster fullerene C60 is a Euler polyedra: It is built around 60 atoms, 32 faces and 90 edges. Check it with Euler’s formula, it does work!
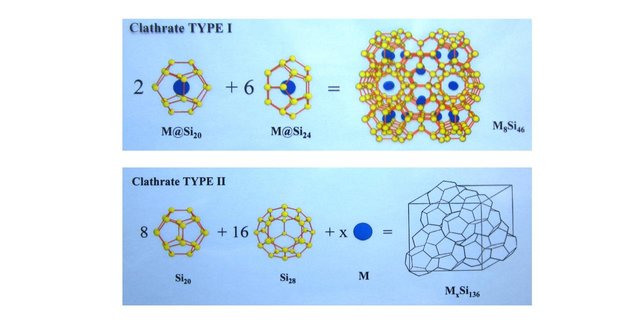
When a sp3 configuration is eclipsed, two structures can emerge built solely on Euler polyhedra:
- The Type 1 Clathrate structure, made of 2 dodecahedra (12 sides, 20 atoms, 30 edges) and 6 tetrakaidecahedrea (14 sides, 274 atoms, 36 edges).
- The Type 2 Clathrate structure, made of 8 dodecahedra (12 sides, 20 atoms, 30 edges) and 16 hexakaidecahedrea (16 sides, 39 atoms, 42 edges).
The images below have been extracted from a presentation I gave years ago on this topic. You can see the two structures with atoms intercalated within the cages.
3/ How do clathrate crystals form ?
Many of you may know that when you put graphite (a 2-dimensional structure made of sheets of carbon) under high pressure and high temperature, the carbon atoms rearrange themselves in order to fill up space optimally: the graphite becomes diamond and is stable. This is how industrial synthetic diamonds are synthesized.

The same thing happens with silicon, the material I was working on during my PhD. Under high pressure high temperature conditions, silicon atoms rearrange themselves and take on a sp3 structure with the tetrahedra in an offset configuration.
We have seen that clathrates are kind of empty structure, with big holes at the center of the cages. So how can we synthesize these materials and avoid ending up with a diamond structure? The trick I used was to ‘frustrate’ the crystal.
I started first by synthesizing a material very unstable in air, sodium silicide, of formula NaSi. It consist of small tetrahedron of silicon separated by sodium atoms as shown in the drawing below.

By applying high pressure and high temperature conditions to the silicide, the silicon atoms try to evolve towards a diamond structure but are blocked by the sodium atoms. They are forced to roll themselves around the sodium atoms, hence taking an eclipsed configuration. In such situation, the presence of other atoms in the initial lattice forces the crystal to take a clathrate structure.
This kind of structural frustration is not uncommon in nature actually. These clathrate structures exist also as certain types of water ice. They are called clathrate hydrates and are massively present at the bottom of our oceans, trapping methane molecules in their cages (this material is also called more commonly gaz hydrates)… This special ice could be melting soon due to global warming and liberate huge amounts of methane in the atmosphere… which in turn would make things worse.. I might talk about this in another post, more oriented toward climate concerns…

With my research team of the time, I carried out many characterization experiments on this new silicon clathrate material and got stunning results. For example by bombarding the material with a neutron beam, and then analyzing the resulting inelastic diffusion of the neutrons within the material, we were able to deduct information about the stiffness of the bonds. This led us to conclude that the clathrate structures of silicon are harder than of the corresponding silicon diamond one. I am very proud of the paper we wrote about this and that was published as a highlight in Physics Review Letters: (https://journals.aps.org/prl/abstract/10.1103/PhysRevLett.83.5290)
It was not long till I started reflecting on trying to make carbon clathrate structures: by extrapolation, such material would the hardest material to exist, harder than diamond, with huge potential applications at the industrial level!
Unfortunately, I never had the opportunity to explore further as I had to leave academia soon after my work on silicon clathrates was completed.

4/ Back to our dying stars and their dazzling surface chemistry
Now, let’s come back to cool white dwarfs… the surface is around 4000K, composed of carbon with a little bit of helium, and maybe traces of oxygen. The surface is cooling while being subject to a huge amount of pressure: consequentely the carbon nuclei plasma is progressively crystallizing into a diamond structure to optimize space.
But… wait…
What about the effect of the presence of helium and oxygen impurities within the carbon plasma? During crystallization, they have no place to go while surrounded by this carbon madness!
Well, these atoms have also the right to exist, and are going to make a stand for it! They will frustrate the formation of the carbon crystal and make the carbon bonds wrap around them. The newly formed sp3 carbon bonds will have as only choice to take on an eclipsed configuration and form clathrate structures!
The crust of cold white dwarfs could be exhibiting a clathrate crystalline structure!
Of course, all depends on the distribution and concentration of the helium and oxygen frustrators present during the crystallization process, however I do envision the possibility of having beautiful crystals He8C46 and / or He8O16Si136 embedded within a diamond matrix.
What a hoot it would be to be able to walk on such a surface. I am really looking forward for the genius that will invent anti-gravity suits ;-) !

5/ Conclusion
The hypothesis presented in this article proposes that when white dwarfs cool down and progressively crystallize, a relatively complex solid state chemistry occurs within its crust. It is plausible that when the carbon plasma freezes, it takes on a diamond structure. It is also plausible that carbon clathrates of various stoichiometry could form due to the presence of helium and oxygen nuclei in this environment.
We probably will not be able to verify this idea within the next few centuries, so… what do you think?
Does this idea make sense?
Feel free to discuss it in the comments!

Sources and references:
- "Les clathrates NaxSi136 et Na8Si46: vers un nouveau silicium" (E. Reny, These de doctorat, Universite Bordeaux I, 1999)
- "High Pressure Behavior of Silicon Clathrates: A New Class of Low Compressibility Materials" (San Miguel et al, Phys. Rev. Lett. 83, 5290 – 1999)
- "Astrophysics High Level" Booklet written for my students)
- "Remarkable White Dwarf Star Possibly Coldest, Dimmest Ever Detected" (National Radio Astronomy Observatory website)
Image credits:
- Comet for the frieze: John Vermette - www.johnsastrophotos.com, CC BY-SA 4.0, (source)
- Asteroid in orbit around a white dwarf: NASA, ESA, STScI, and G. Bacon (STScI) (source)
- Tetrahedra and clathrate illustrations, picture of the high pressure high temperature press: @muphy
- NaSi structure extracted from my PhD thesis (source)
All other pictures (all found with Google image advanced with license for modification and commercial use)

Hi,
I’m @muphy, My life revolves around music production, teaching sciences, and discovery through travel.
You enjoyed that post? Resteem and Upvote!
You are interested in these topics? Follow me!
Awesome topic Muphy! i did some research on Methane Clathrates a few years back when said sources suspected that might be the cause of the Bermuda Triangle. I found it highly probable as both buoyancy and the density of air are affected by methane release. I had a question and correct me if im wrong, but dont stars create elements all the way up to lithium in their process of burning out and dying?
Hi @csusbgeochem1, you worked on Gas Hydrates! so you must be familiar with the clathrate structure! Cool, not many people are :-).
Well all the periodic table comes from stars. Hydrogene, Helium and traces of Lithium were formed during the primordial nucleosynthesis (The synthesis of the first atoms after the big bang).
That provided the material to make up stars. Depending on their mass, these stars will create the other elements. If they are very small, up to Helium, for small to medium sized like the sun, up to oxygen, and for bigger ones, those destined to explode in supernovas, up to Iron.
At the end of their lives, the bigger stars will produce all the other elements during a supernovae phase. In the future, I'll probably write a much more detailed post about this fascinating phenomena describing the various fusion reactions... These are not always simple and straight forward, but nature always finds its own ways :-)
This is amazing!
Why did you have to leave academia/ any plans to return?
Thank you @mountainwashere. You know the proverb, when there are budget cuts, last in, first out. Well, I wasn't even in yet when the official position I was going to take was cancelled after huge cuts had been decided in French public research. I even had an office being set for me by my future colleagues and a million francs accepted to buy the tools I needed for my research (A high pressure high temperature press).
So after my PhD in France and some post doc research in Japan, the next step never happened, and I became like many other promising young researchers: looking for a job to get money and put food on the table. I turned to the industry...
After a decade, bored and disheartened by the hypocrisy and lack of vision of this sector, I left and created my own company. Best decision ever: now I am doing multiple activities that used to be hobbies and it is working very well. And now, there is Steemit that just added itself to the list of fun activities ;-)!
No plan to return... If I was offered a position, I would not settle for less than research director in order for me to carry out the research projects I want, and decide of the research directions , have a team, and mentor / guide a bunch of PhD students. Unfortunately, the French research system is really not the most flexible for this kind of thing, haha! So this will most probably never happen!
Ahh, that's too bad. Happy you're doing something you love now instead, though!
#mindblown by this article. Thanks :)
Anyways sir @murphy, this is something not related by your post and if you don't mind me asking for your recommendations. I'm from Philippines and i'm planning to pursue Medicine and its been a while since i graduated from college in bachelor's degree, can i ask for recommendations on what to review in Physics? Generally concept and understanding of Physics. Thanks.
Have a great day ahead.
Hello @iamkuyaj, thank you for your kind words.
If you are going for medecine studies, as far as Physics is concerned, I would look into medical physics. The principles behind echography, NMR, radiographyetc. i.e. the various scans that exist. That means also being familiar with nuclear decay concepts (half life) and certain aspects of particle physics (positron beam generation and its effect on the body, pi meson radiation therapy etc...).
The best for you would be to contact the Uni you wish to enter, or visit their website, and ask them or check the details of the course you wish to follow. That should tell you what you need to prepare in all fields.
Be well, and good luck!
thank you Dr. @murphy :)
i'm already familiar with nuclear decay concepts. Just need to review it. Thanks for the advice.
Anti-gravity suits are not for tomorrow, unfortunately ;)
Oh you never know, lol!
The discoveries in fundamental sciences made these past few decades have been more fascinating than the most creative of science fiction!
So even though what we know today strictly forbids such a suit... I enjoy keeping my mind open to the wildest ideas ;-)
Me too. But here, we may need to wait a couple of years, at least :D
Great article with lots of interesting information my mind is truly blown away wonderful post by you :)
Thank you! I am glad you enjoyed reading it.
Someone should really invent that anti-gravity suit! :)
excellent post friend voted greetings ..
Thank you
good work nice post thanks for info
Thank you
aah good work
appreciate