Revolutionary Treatment For Cancer Tested on Humans
Chinese scientists injected for the first time into a man cells that contained edited genes using the technique CRISPR-Cas9, test that could trigger a duel with their US colleagues.
Foto: Gulliver/Getty Images
On 28 October, a team led by oncologist Lu You of Sichuan University in Chengdu injected the modified cells in a patient with aggressive lung cancer, in a clinical trial conducted in a local hospital.
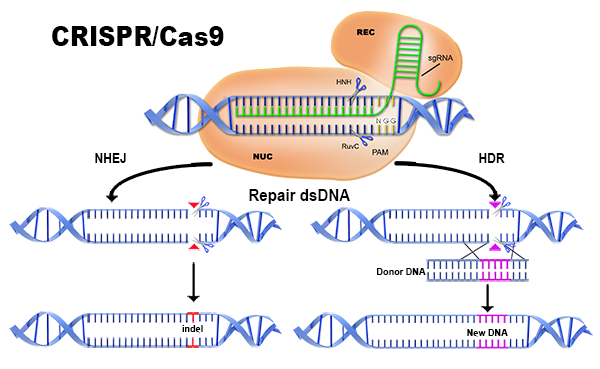
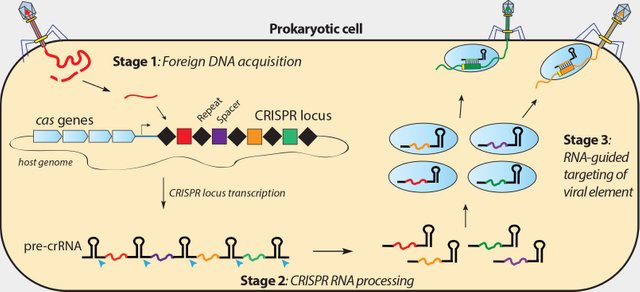
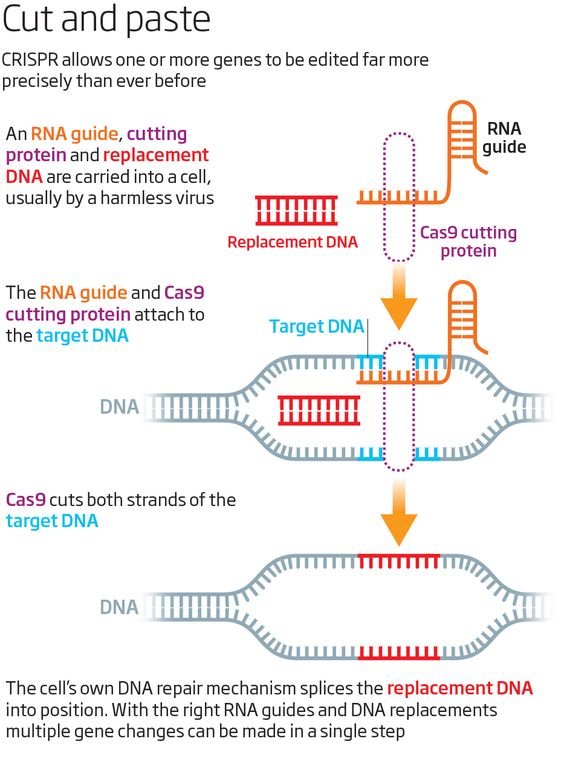
The researchers extracted immune cells from the individual's blood injected and then disabled a gene in them using the technique CRISPR-Cas9, combining an enzyme cutting of DNA with a guide molecular which can be programmed to instruct enzyme exactly where to cut. The removed genetic codes are PD-1 protein, which usually hinder the immune response of the cell,in advantage for cancer to proliferate.
Previous clinical trials have used edited cells in different technique that enchanted doctors. " Entering CRISPR, which is simpler and more effective than other techniques will likely accelerate the race to bring the use of cell edited genetic clinics worldwide", said Carl June, which specializes in immunotherapy at the University of Pennsylvania in Philadelphia who led previous studies.
"I think this will trigger 'Sputnik 2.0', a duel biomedical progress in the field between China and the US, which is important because competition usually improve the final product," said Carl June
Carl June is a scientific advisor for the planned US clinical trial that will use the technique to target CRISPR three genes in cells participants, in order to treat various cancers. He argues that clinical testing could begin in the first half of 2017.
On the other hand, in March 2017 a group from the University of Beijing hopes to start clinical trials using the technique three CRISPR against cancer cells in the bladder, prostate and kidney, but they have not yet approved the financing.
The clinical trial headed by Lu You received ethical approval from a board assessment in July. Injection participants would start in August but the date was then postponed, says Lu You, because crop production and amplifying cells took longer than anticipated.
You Lu's team conducted a edited cell culture, increasing their number, and injected them back into the patient, suffering from metastatic lung cancer.
The coordinator maintains that the treatment went well and the participant will receive a second injection, but refused to provide more details. The team plans to treat a total of ten people who will each receive two, three or four injections.
It is primarily a safety test, and participants will be monitored for six months to determine whether injections cause serious side effects. Team doctor Lu You will however monitor patients after this period to find out if the treatment benefic.
And other oncologists are satisfied with the entry CRISPR stage cancer treatment. "The technology able to do this is incredible," says Rizvi Naiyer of the Columbia University Medical Center in New York. Antonio Russo of the University of Palermo in Italy notes that PD-1 neutralizing antibodies were able to stop cancer, which bodes well for attacking technique CRISPR protein. "It is a formidable strategy. The logic is strong," he said.
Rizvi Naiyer but questions whether the current clinical trial will succeed. Extraction process, genetic modification and multiplication of cells is "a huge task and not very easy to achieve," he said.
'If you do not show a significant increase in efficiency will be difficult to justify a continuation" said the researcher. He doubts that the technique will be one superior to the use of antibodies that can be propagated in unlimited quantities in clinics. You Lu says the problem is evaluated in the clinical trial, but it is too early to say which approach is better.
References:
- http://www.nature.com/nbt/journal/v33/n4/full/nbt.3155.html
- http://www.nature.com/news/crispr-gene-editing-tested-in-a-person-for-the-first-time-1.20988
- https://en.wikipedia.org/wiki/CRISPR
Join us on #steemSTEM
Very nice post detailing new and cutting edge applications of the CRISPR/Cas9 gene editing technology! Will be very exciting to see how this tech is applied both in this context and in the new and exciting ways that are sure to follow.
As a bonus, and in addition to resteeming for exposure. We are awarding you a small 5 Steem Power deposit as a thank you for creating quality STEM related postings on Steemit. We hope you will continue to educate us all!
Thank you! I really appreciate how this project aims to boost the steemians.
It is always great to read about cancer treatment. In my opinion, this is one of the greatest challenge in medicine today.
This is an interesting application of Cas9, creating a few cells with a specific phenotype, that can then be administered to a patient.
yes, the future is creating cells with specific actions like these
Hope in the future will be a real succes for all
If there will be a serious funding for these tests I am sure we will have the treatment