Chemistry Basics

In germany, classes on chemistry usually start with explaining a few chemical concepts, then showing some chemical reactions (that is the change of one substance into another by applying heat). Good classes even allow pupils to do this themselves, teaching how to use a bunsen burner , how it functions, and where the hottest point of the flame is. This is important knowledge for all further practical sessions, as errors can lead to wrong results or, in the worst case, minor accidents.
The Bunsen Burner
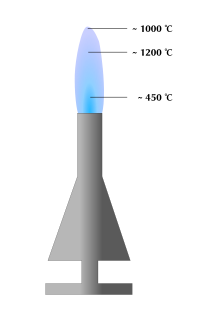
Info: What happens
The reason is that, due to missing oxygen, the gas (usually methane) is burned imperfectly. Instead of the whole gas turning into carbon dioxide and water through burning, the gas is stripped of its hydrogen-atoms leaving begind the naked carbon atoms, which are emitting the orange-yellow light at high temperature. As the carbon reaches the edge of the flame, it is turned into carbon dioxide as well. This can be confirmed by putting something inflammable into the orange flame for a moment, and pulling it out. Soot will have collected on it. In the flame with extra air, the carbon can react directly into carbon dioxide, resulting in the loss of the glow, a higher temperature, and in not leaving any soot on things put into the flame.
Pure Substance and Substance Mixtures
These are the first two categories taught in school. There are pure substances (like pure water, iron, or even sand) and substance mixtures (like milk, smoke, or water with salt dissolved in it). Pure substances have precicely defined properties that can be used to identify them. For example Water usually boils at 100 °C, is a liquid, is color- and tasteless, and freezes at 0 °C. On the other hand there are substance mixtures - substances with two ore more components in them. Oftentimes the substances contained within a substance mixture can be seperated by exploiting the properties of the components.
Homogenous Mixtures
These mixtures are uniform mixtures. (Homo is derived from the greek homos, meaning identical, or same.) You can't even make out any different components by looking closely. Everything blends neatly into one mixture. An easy example for that is dissolving salt into water. You can taste the difference, but you can't see it. And yet, there is a possibility that allows these two components to be seperated: Just boil off the water.
Experiment
If you want to try this at home, then there is an easy way to do this. You need water, salt, a clasp from a champagne bottle and a tea candle. Dissolve a tea spoon of salt in half a glass of water. Then remove any foil from the clasp, then turn the small "dish" that held the cork in the bottle upside down. Put all of this over the tea candle, fill some water in the "dish" (just a few droplets), and lit the candle. After a few minutes the water will have evaporated, leaving behind a white crust: the salt.
If you use tap water, you can do it with just the water all the same, and you'll probably be left with a white crust as well, just a lot less, as there is no pure water coming from our taps. It may also have different colors, depending on where you live and what water you have.
Heterogenous Mixtures
These mixtures are not uniform. In fact, using a microscope you can make out the components with the naked eye. A well known heterogenous mixture is milk. It is a mixture of fat in water (and fat and water don't mix). Even homogenized milk is still a heterogenous mixture. The only thing that is made uniform is the size of the fat-droplets within the milk, keeping the emulsion stable for a longer time. An easy way to seperate the water from the fat in milk is to add salt. The salt will pull out the water, and the fat will congeal on top of the milk. The result is not pretty, but if you want to try, be my guest.
Is seperating Mixtures Chemistry?
Yes, and no. There is no chemical reaction taking place, in this sense, it is not chemistry. On the other hand, seperating substances is a necessary skill for any chemist to know, so it is still part of chemistry.
What can I do with this knowledge?
Look on the many things you buy: Food, face creams, medicine ... Those are all mixtures. You can even try the salt trick on sun screen. Or face creams. Some may take a bit longer than others to seperate, but everything that contains water and fat-like substances can be seperated by adding salt. But here is a small warning: After doing so you might not want to use those products anymore ... The result is not appealing.
Congratulations @lunanightshade! You received a personal award!
Click here to view your Board
Congratulations @lunanightshade! You received a personal award!
You can view your badges on your Steem Board and compare to others on the Steem Ranking
Vote for @Steemitboard as a witness to get one more award and increased upvotes!